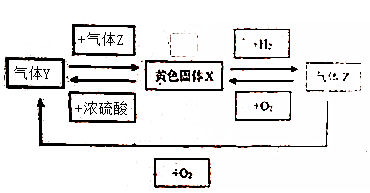
��Ŀ����
16��ij��ѧ��ȤС������NaI��Һ��ͨ������Cl2�õ������Һ�������ú����Һ��ȡNaI���壬ʵ���������£�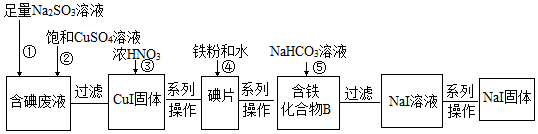
��֪��Ӧ�ڣ�2I-+2Cu2++SO32-+H2O�T2CuI��+SO42-+2H+��
�ش��������⣺
��1������ʵ������Ҫ�IJ�����������ͨ©�����ձ�����������
��2����Ӧ����CuI����������ԭ����ֻ��NO2���÷�Ӧ�Ļ�ѧ����ʽΪ2CuI+8HNO3��Ũ���T2Cu��NO3��2+4NO2��+I2+4H2O��
����95.5g CuI���뷴Ӧ������Ҫ�����5.6L��O2���ܽ����ɵ�NO2��ˮ����ȫת��Ϊ���ᣮ
��3��������B�к�����Ԫ�أ���Ԫ��ԭ������һ��Ԫ��ԭ�ӵ����ʵ���֮��Ϊ3��8������B�Ļ�ѧʽΪFe3I8��
��4����Ӧ�������ɺ�ɫ�������ɫ���壬��ɫ������׳�Ϊ��������������Ӧ�ݵĻ�ѧ����ʽΪFe3I8+8NaHCO3�TFe3O4+8NaI+8CO2��+4H2O��
��5����������Cl2ͨ�뺬12g NaI����Һ�У�һ��ʱ������Һ���ɵõ����������Ϊ4.68g��
���� �����Һ�����������ƺͱ�������ͭ��Һ������������ԭ��Ӧ����CuI���壬����Ũ���ᣬ�ɵõ��⣬Ȼ��������ۺ�ˮ���õ�����������B��������B�к�����Ԫ�أ���Ԫ��ԭ������һ��Ԫ��ԭ�ӵ����ʵ���֮��Ϊ3��8����ӦΪFe3I8������̼��������Һ�ɵõ�NaI��Һ��ͬʱ�õ���ɫ����Fe3O4������Fe3I8+8NaHCO3�TFe3O4+8NaI+8CO2��+4H2O����Һ���������ᾧ�õ�NaI���壬�Դ˽����⣮
��� �⣺��1������ʵ������Ҫ�IJ�����������ͨ©�����ձ����������������������������ʴ�Ϊ����������
��2����Ӧ����CuI����������ԭ����ֻ��NO2���÷�Ӧ�Ļ�ѧ����ʽΪ2CuI+8HNO3��Ũ���T2Cu��NO3��2+4NO2��+I2+4H2O������95.5g CuI���뷴Ӧ����n��CuI��=$\frac{95.5g}{191g/mol}$=0.5mol���ɷ���ʽ��֪����1molNO2����ͨ����������4NO2+2H2O+O2=4HNO3������0.25mol���������Ϊ5.6L��
�ʴ�Ϊ��2CuI+8HNO3��Ũ���T2Cu��NO3��2+4NO2��+I2+4H2O��5.6��
��3��������B�к�����Ԫ�أ���Ԫ��ԭ������һ��Ԫ��ԭ�ӵ����ʵ���֮��Ϊ3��8����һ��Ԫ��ΪI������B�Ļ�ѧʽΪFe3I8���ʴ�Ϊ��Fe3I8��
��4����Ӧ�������ɺ�ɫ�������ɫ���壬��ɫ������׳�Ϊ������������ӦΪ��������������ɫ����Ϊ������̼����Ӧ�ݵĻ�ѧ����ʽΪFe3I8+8NaHCO3�TFe3O4+8NaI+8CO2��+4H2O��
�ʴ�Ϊ��Fe3I8+8NaHCO3�TFe3O4+8NaI+8CO2��+4H2O��
��5��n��NaI��=$\frac{12g}{150g/mol}$=0.08mol����������������Ӧ����0.08molNaCl����m��NaCl��=0.08mol��58.5g/mol=4.68g��
�ʴ�Ϊ��4.68��
���� ���⿼�����ʵ��Ʊ���������ѧ���ķ���������ʵ�������Ŀ��飬Ϊ�߿��������ͺ�Ƶ���㣬ע�����ʵ������̺��Ʊ�ԭ�����Ѷ��еȣ�

 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�| M | N | ||
| X | Y | Z |
| A�� | �⻯����ȶ��ԣ�N��Z������Ϊ���⻯����ӿ��γ���� | |
| B�� | Ԫ�صķǽ����ԣ�M��Y������Ϊ����M����YN2��Ӧ���ɵ���Y | |
| C�� | �۷е㣺YN2��MN2������ΪYN2�ķ��Ӽ�������ǿ | |
| D�� | �����ӵİ뾶��N��X������Ϊ���Ǿ�����ͬ�ĵ��Ӳ�ṹ��X�ĺ˵�������� |
| ѡ�� | �������ʵ | ���� |
| A | ����������ɱ������ | �����Ļ�ԭ��ʹϸ���ĵ����ʱ��� |
| B | ������������ȱ����ƶѪ | �����������������Ȼ��� |
| C | ������������������еĹ����� | ����������ǿ��������������������̼ |
| D | �ý������Ը�����صĹ�������ˮ�����ʼ� | �������������ˮ���ͷŵĴ������ϩ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | ����������Һ��ͨ������ Fe2++Cl2�TFe3++Cl- | |
| B�� | ̼��þ��ϡ���ᷴӦ��MgCO3+2H+�TMg2++H2O+CO2�� | |
| C�� | ʯ������ϡ���ᷴӦOH-+H+�T2H2O | |
| D�� | ϡ�������ͭƬ�ϣ�Cu+2H+�TCu2++H2�� |
| A�� | ������AB2�����������ӻ����� | |
| B�� | ������AB2�ľ�������Ƿ��Ӿ��塢���Ӿ����ԭ�Ӿ��� | |
| C�� | ������AB2���۵㲻���ܸܺ� | |
| D�� | ������AB2�Ŀռ�����ṹ��������V�νṹ |
| A�� | ��CuSO4��Һ�͡���һ���ʡ���CuH�ķ�Ӧ�У���������ԭ�۵�������⡰��һ���ʡ��ڷ�Ӧ���������� | |
| B�� | CuH��������ȼ�յĻ�ѧ��Ӧ����ʽΪ2CuH+3Cl2 $\frac{\underline{\;��ȼ\;}}{\;}$2CuCl2+2HCl | |
| C�� | CuH�ܽ���ϡ���������ɵ����������� | |
| D�� | ���ѡ��C��Ӧ�������˱�״����22.4 L�����壬��ת�Ƶ���3 mol |