��Ŀ����
3��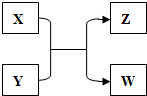 X��Y��Z��W���������������ת����ϵ������X��WΪ���ʣ�Y��ZΪ�����δ�г���Ӧ��������
X��Y��Z��W���������������ת����ϵ������X��WΪ���ʣ�Y��ZΪ�����δ�г���Ӧ����������1����Z�������г��õĵ�ζƷ��W��������Һ��������X�����ڳ����µ���ɫΪ����ɫ��
��2����X�dz������ܶ���С�����壬Y�����ַǽ���Ԫ����ɣ�����һ���ڵؿ��еĺ���������������һ���ں�ˮ�еĺ������������������Z�Ļ�ѧʽΪHCl��
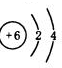
��3����Xԭ�ӵ���������Wԭ����������2������Xԭ������������Ϊ2����W��ԭ�ӽṹʾ��ͼΪ
 ��
��
���� ��1����Z�������г��õĵ�ζƷ��W��������Һ��������WΪI2��ZΪNaCl��XΪ������YΪNaI��
��2����X�dz������ܶ���С�����壬��XΪ������Y�����ַǽ���Ԫ����ɣ�����һ���ڵؿ��еĺ���������������һ���ں�ˮ�еĺ������������������YΪSiCl4��WΪSi��ZΪHCl��
��3����Xԭ�ӵ���������Wԭ����������2������Xԭ������������Ϊ2����XΪMg��YΪCO2��WΪC��ZΪMgO��
��� �⣺��1����Z�������г��õĵ�ζƷ��W��������Һ��������WΪI2��ZΪNaCl��XΪ������YΪNaI��X�����ڳ����µ���ɫΪ������ɫ���ʴ�Ϊ������ɫ��
��2����X�dz������ܶ���С�����壬��XΪ������Y�����ַǽ���Ԫ����ɣ�����һ���ڵؿ��еĺ���������������һ���ں�ˮ�еĺ������������������YΪSiCl4��WΪSi��ZΪHCl���ʴ�Ϊ��HCl��
��3����Xԭ�ӵ���������Wԭ����������2������Xԭ������������Ϊ2����XΪMg��YΪCO2��WΪC��ZΪMgO��W��ԭ�ӽṹʾ��ͼΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���� ���⿼�������ƶϣ���Ҫѧ����������Ԫ�ػ�����֪ʶ��ע��Mg�������̼��Ӧ���Ѷ��еȣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
13���������ŷḻ�ĺ�ˮ��Դ������������Դ�ͱ�����������ʡ�ɳ�����չ����Ҫ��֤��
��1����ˮ������������Եõ���ˮ�Ȼ�þ����ˮ�Ȼ�þ�ǹ�ҵ��ȡþ��ԭ�ϣ���д������ˮ�Ȼ�þ��ȡ����þ�Ļ�ѧ��Ӧ����ʽMgCl2 $\frac{\underline{\;���\;}}{\;}$Mg+Cl2����
��2��ij���������������л��������Cu2+��Pb2+����ˮ���ŷ�ǰ���ó�������ȥ���������ӣ������������ݣ�����ΪͶ��Na2S��ѡ�Na2S����NaOH����Ч�����ã�
��3�����������ڽ��յ���Դ������ռ�ϴ���أ������ŷų���SO2�����һϵ�л�������̬���⣮���ú�ˮ������һ����Ч�ķ������乤��������ͼ��ʾ��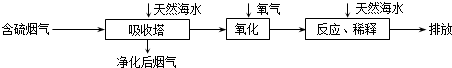
����Ȼ��ˮ��pH��8��ˮ����Ҫ����Na+��K+��Ca2+��Mg2+��Cl-��SO42-��Br-��CO32-��HCO3-�����ӣ��������ӷ���ʽ������Ȼ��ˮ�������Ե�ԭ��CO32-+H2O?HCO3-+OH-�� HCO3-+H2O?H2CO3+OH-����дһ������
��ij�о�С��Ϊ̽����ߺ���������SO2������Ч�ʵĴ�ʩ����������Ȼ��ˮ���պ���������ģ��ʵ�飬ʵ������ͼ��ʾ��

�������ͼʾʵ��������������һ��Ũ�Ⱥ���������SO2������Ч�ʣ����һ�����������飺���ͺ��������¶ȣ������٣���
����Ȼ��ˮ�����˺��������������H2SO3��HSO3-�ȷ��ӻ����ӣ�ͨ�����������Ļ�ѧԭ����2H2SO3+O2=4H++2SO42-��2HSO3-+O2=2H++2SO42-����дһ����ѧ����ʽ�����ӷ���ʽ����������ġ���ˮ����Ҫ�����������Ȼ��ˮ��֮��Ϻ�����ŷţ��ò�������ҪĿ�����к͡�ϡ�;�����������ˮ�����ɵ��ᣨH+����
��1����ˮ������������Եõ���ˮ�Ȼ�þ����ˮ�Ȼ�þ�ǹ�ҵ��ȡþ��ԭ�ϣ���д������ˮ�Ȼ�þ��ȡ����þ�Ļ�ѧ��Ӧ����ʽMgCl2 $\frac{\underline{\;���\;}}{\;}$Mg+Cl2����
��2��ij���������������л��������Cu2+��Pb2+����ˮ���ŷ�ǰ���ó�������ȥ���������ӣ������������ݣ�����ΪͶ��Na2S��ѡ�Na2S����NaOH����Ч�����ã�
| ���ܵ���� | Cu��OH��2 | CuS | Pb��OH��2 | PbS |
| Ksp | 4.8��10-20 | 6.3��10-36 | 1.2��10-15 | 1.0��10-28 |

����Ȼ��ˮ��pH��8��ˮ����Ҫ����Na+��K+��Ca2+��Mg2+��Cl-��SO42-��Br-��CO32-��HCO3-�����ӣ��������ӷ���ʽ������Ȼ��ˮ�������Ե�ԭ��CO32-+H2O?HCO3-+OH-�� HCO3-+H2O?H2CO3+OH-����дһ������
��ij�о�С��Ϊ̽����ߺ���������SO2������Ч�ʵĴ�ʩ����������Ȼ��ˮ���պ���������ģ��ʵ�飬ʵ������ͼ��ʾ��

�������ͼʾʵ��������������һ��Ũ�Ⱥ���������SO2������Ч�ʣ����һ�����������飺���ͺ��������¶ȣ������٣���
����Ȼ��ˮ�����˺��������������H2SO3��HSO3-�ȷ��ӻ����ӣ�ͨ�����������Ļ�ѧԭ����2H2SO3+O2=4H++2SO42-��2HSO3-+O2=2H++2SO42-����дһ����ѧ����ʽ�����ӷ���ʽ����������ġ���ˮ����Ҫ�����������Ȼ��ˮ��֮��Ϻ�����ŷţ��ò�������ҪĿ�����к͡�ϡ�;�����������ˮ�����ɵ��ᣨH+����
14�������йػ�ѧ�����ʾ��ȷ���ǣ�������
| A�� | HCl�ĵ���ʽ��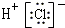 | B�� | ${\;}_{8}^{18}$O2-���ӵ�ԭ�ӽṹʾ��ͼ�� | ||
| C�� | CO2���ӵĽṹʽ��O=C=O | D�� | ���Ȼ�̼���ӱ���ģ�ͣ� |
11��2015��8��12������Σ��Ʒ�ֿⷢ���ı�ը�¹ʣ��ٴξ�ʾ���Dz��ɺ��ӻ�ѧ�Լ��Ĺ淶���森������������ȷ���ǣ�������
| A�� | �Ʊ�����ϸ��ƿ�в���ú��Һ�� | |
| B�� | ������ˮ��������ɫƿ�в������䰵�� | |
| C�� | NaOH��Һ��������ɫ�Լ�ƿ�в�Ҫ�������� | |
| D�� | ����������Һ����ڼ����������۵��Լ�ƿ�� |
18��ijͬѧ����ʵ��֤������ͭ��Һ����ɫ��SO42-�أ�������ʵ����������ǣ�������
| A�� | �۲���������Һû����ɫ | |
| B�� | ��ˮϡ������ͭ��Һ����Һ��ɫ��dz | |
| C�� | ������ͭ��Һ�еμ��������Ȼ�����Һ�����ã���Һ��ɫδ��ʧ | |
| D�� | ������ͭ��Һ�еμ�����������������Һ�����ã���Һ��ɫ��ʧ |
8����������Ϊ������ƿ�����ǣ�������
| A�� |  | B�� |  | C�� |  | D�� |  |
15����Ԫ�������ڱ��е�λ���ǣ�������
| A�� | �ڶ��ڢ�A�� | B�� | �����ڢ�A�� | C�� | �ڶ��ڢ�A�� | D�� | �����ڢ�A�� |
12�������йػ�ѧҩƷ�Ĵ��˵������ȷ���ǣ�������
| A�� | Һ���ӷ���ʵ���ҳ���ˮҺ�Ⲣ�ܷⱣ�� | |
| B�� | ��������������������Ӧ����������ú���� | |
| C�� | Ũ������ǿ��ʴ�ԣ�����ʱ������������������ | |
| D�� | ��������ҺӦ�ô�������ϸ��ƿ���� |
11�������ֽ�����ɵĻ����4.8g����������ȫ��Ӧʱ����������2.24L����״���������������ɿ����ǣ�������
| A�� | þ���� | B�� | �ƺ��� | C�� | �ƺ��� | D�� | ����ͭ |