��Ŀ����
��13.6gCu��Cu2O�Ļ�����м���ijŨ�ȵ�ϡ����0.25L������������ȫ��Ӧ������NO��Cu��NO3��2����������Һ�м���0.5mol/L��NaOH��Һ1L����Ӧ����Һǡ�ó����ԣ�������������ȫ��������������Ϊ19.6g������˵������ȷ���ǣ�������
| A��Cu��Cu2O�����ʵ���֮��Ϊ2��1 |
| B����������ʵ���Ũ��Ϊ2 mol/L |
| C��Cu��Cu2O�����ᷴӦ��ʣ��HNO3�����ʵ���Ϊ0.5mol |
| D��������NO�ڱ�״���µ����Ϊ2.24L |
���㣺�йػ���ﷴӦ�ļ���
ר�⣺�غ㷨
��������������Һ�м���0.5mol/L ��NaOH��Һ1L����Һ�����ԣ�������������ȫ��������ʱ��Һ������ΪNaNO3��n��NaNO3��=n��NaOH��=0.5mol/L��1L=0.5ml������ΪCu��OH��2������Ϊ19.6g�������ʵ���Ϊ��
=0.2mol������ͭԪ���غ���n��Cu��+2n��Cu2O��=n[Cu��OH��2]�����Է�Ӧ�����Һ��n[[Cu��NO3��2]=n[Cu��OH��2]=0.2mol��
A����Cu��Cu2O�����ʵ����ֱ�Ϊxmol��ymol�����ݶ�����������ϵʽn��Cu��+2n��Cu2O��=n[Cu��OH��2]���з��̼��㣬Ȼ������Cu��O�����ʵ���֮�ȣ�
B������NԪ���غ��֪ԭ������Һn��HNO3��=n��NO��+n��NaNO3��������A�����Cu��Cu2O�����ʵ��������õ���ת���غ����n��NO��������NaԪ�ؿ�֪n��NaNO3��=n��NaOH�����ٸ���c=
���㣻
C����Ӧ�����Һ�м�������������Һ����������������ͭ��Ӧ��ʣ����������������ᷴӦ�����Ϊ��������Һ�����ݵ�Ԫ���غ��֪��Ӧ����Һ��n��HNO3��+2n[[Cu��NO3��2]=n��NaNO3�����ݴ˼��㣻
D������A�����Cu��Cu2O�����ʵ��������õ���ת���غ����n��NO�����ٸ���V=nVm����NO�������
| 19.6g |
| 98g/mol |
A����Cu��Cu2O�����ʵ����ֱ�Ϊxmol��ymol�����ݶ�����������ϵʽn��Cu��+2n��Cu2O��=n[Cu��OH��2]���з��̼��㣬Ȼ������Cu��O�����ʵ���֮�ȣ�
B������NԪ���غ��֪ԭ������Һn��HNO3��=n��NO��+n��NaNO3��������A�����Cu��Cu2O�����ʵ��������õ���ת���غ����n��NO��������NaԪ�ؿ�֪n��NaNO3��=n��NaOH�����ٸ���c=
| n |
| V |
C����Ӧ�����Һ�м�������������Һ����������������ͭ��Ӧ��ʣ����������������ᷴӦ�����Ϊ��������Һ�����ݵ�Ԫ���غ��֪��Ӧ����Һ��n��HNO3��+2n[[Cu��NO3��2]=n��NaNO3�����ݴ˼��㣻
D������A�����Cu��Cu2O�����ʵ��������õ���ת���غ����n��NO�����ٸ���V=nVm����NO�������
���
�⣺��������Һ�м���0.5mol/L ��NaOH��Һ1L����Һ�����ԣ�������������ȫ��������ʱ��Һ������ΪNaNO3��n��NaNO3��=n��NaOH��=0.5mol/L��1L=0.5ml������ΪCu��OH��2������Ϊ19.6g�������ʵ���Ϊ��
=0.2mol������ͭԪ���غ���n��Cu��+2n��Cu2O��=n[Cu��OH��2]�����Է�Ӧ�����Һ��n[[Cu��NO3��2]=n[Cu��OH��2]=0.2mol��
A����Cu��Cu2O�����ʵ����ֱ�Ϊxmol��ymol�����ݶ��������� 64x+144y=13.6������ͭԪ���غ���x+2y=0.2���������̽��x=0.1��y=0.05����n��Cu����n��Cu2O��=0.1mol��0.05mol=2��1����A��ȷ��
B������NԪ���غ��֪n��HNO3��=n��NO��+n��NaNO3�������ݵ���ת���غ��֪��3n��NO��=2n��Cu��+2n��Cu2O��������3n��NO��=2��0.1mol+2��0.05mol�����n��NO��=0.1mol������NaԪ�ؿ�֪n��NaNO3��=n��NaOH��=0.5mol/L��1L=0.5ml������n��HNO3��=n��NO��+n��NaNO3��=0.1mol+0.5mol=0.6mol������ԭ������Һ��Ũ��Ϊ��
=2.4mol/L����B����
C����Ӧ�����Һ�м�������������Һ����������������ͭ��Ӧ��ʣ����������������ᷴӦ�����Ϊ��������Һ�����ݵ�Ԫ���غ��֪��Ӧ����Һ��n��HNO3��+2n[[Cu��NO3��2]=n��NaNO3��������n��HNO3��=n��NaNO3��-2n[[Cu��NO3��2]=0.5mol-2��0.2mol=0.1mol����C����
D����B�м����֪n��NO��=0.1mol�����Ա�״����NO�����Ϊ��0.1mol��22.4L/mol��2.24L����D��ȷ��
��ѡBC��
| 19.6g |
| 98g/mol |
A����Cu��Cu2O�����ʵ����ֱ�Ϊxmol��ymol�����ݶ��������� 64x+144y=13.6������ͭԪ���غ���x+2y=0.2���������̽��x=0.1��y=0.05����n��Cu����n��Cu2O��=0.1mol��0.05mol=2��1����A��ȷ��
B������NԪ���غ��֪n��HNO3��=n��NO��+n��NaNO3�������ݵ���ת���غ��֪��3n��NO��=2n��Cu��+2n��Cu2O��������3n��NO��=2��0.1mol+2��0.05mol�����n��NO��=0.1mol������NaԪ�ؿ�֪n��NaNO3��=n��NaOH��=0.5mol/L��1L=0.5ml������n��HNO3��=n��NO��+n��NaNO3��=0.1mol+0.5mol=0.6mol������ԭ������Һ��Ũ��Ϊ��
| 0.6mol |
| 0.25L |
C����Ӧ�����Һ�м�������������Һ����������������ͭ��Ӧ��ʣ����������������ᷴӦ�����Ϊ��������Һ�����ݵ�Ԫ���غ��֪��Ӧ����Һ��n��HNO3��+2n[[Cu��NO3��2]=n��NaNO3��������n��HNO3��=n��NaNO3��-2n[[Cu��NO3��2]=0.5mol-2��0.2mol=0.1mol����C����
D����B�м����֪n��NO��=0.1mol�����Ա�״����NO�����Ϊ��0.1mol��22.4L/mol��2.24L����D��ȷ��
��ѡBC��
���������⿼��ͭ���仯����֪ʶ��������ԭ��Ӧ�ļ��㣬��Ŀ�ѶȽϴ���ȷ��Ӧ���̡���������غ�����ǽ���Ĺؼ�������������ѧ���ķ�����������������ѧ����������

��ϰ��ϵ�д�
�����Ŀ
���л�ѧ���������ȷ���ǣ�������
A�������ӵĽṹʾ��ͼ 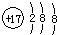 |
| B��HClO��Cl�Ļ��ϼ�Ϊ��+1 |
| C���Ȼ��ƵĻ�ѧʽ��CaCl |
| D������ĵ��뷽��ʽ��H2SO4=H2++SO42- |
������ʵ��װ����ɶ�Ӧ��ʵ�飨����������ʡ�ԣ����ܴﵽʵ��Ŀ���ǣ�������
A�� �Ʊ����ռ���Ȳ |
B�� ��ȥ�Ҵ������е��������� |
C�� �Ʊ������������� |
D�� ʯ�͵ķ��� |
Һ��ʽǦ�����Կ����Եļ�����Ǧ[��CH3SO3��2Pb]����������Ϊ������Һ���õ�س�ŵ���ܷ�ӦΪ
2Pb2++2H2O
Pb+PbO2+4H+������˵����ȷ���ǣ�������
2Pb2++2H2O
| ��� |
| �ŵ� |
| A���õ�طŵ�ʱ���������������� |
| B���ŵ�ʱ�������ĵ缫��ӦʽΪPbO2+4H++2e-�TPb2++2H2O |
| C�����ʱ����Һ��Pb2+�������ƶ� |
| D�����ʱ��������Χ��Һ��pH���� |
��MgCl2��AgCl3�Ļ����Һ�У�Cl-�����ʵ���Ϊ0.7mol��Al3+��Mg2+�����ʵ���֮��Ϊ1��2�������Һ������100mL 8mol?L-1 NaOH ��Һ��ʹ֮��ַ�Ӧ��������������ȷ���ǣ�������
| A����Һ��MgCl2�����ʵ���Ϊ0.2mol |
| B����87.5mL NaOH������Һʱ����������� |
| C��������90.0mL NaOH��Һʱ����Һ��NaAlO2��NaCl�����ʵ���֮��Ϊ2��35 |
| D����100mL NaOH��Һ�պ���ȫ����ʱ��Al3+ ǡ����ȫת��ΪAlO2- |
����ʵ������ܹ��ﵽĿ���ǣ�������
| A����ij��Һ����μ���ϡ���ᣬ���ְ�ɫ��״�������ټ�������NaOH��Һ��������ʧ��˵��ԭ��Һ�д���AlO2- |
| B����ij��Һ�еμ�KSCN��Һʱ���ɫ��˵����Һ�в�����Fe2+ |
| C���ü���ʼ���Fe��OH��3�����Fe��SCN��3��Һ |
| D���ò�˿պȡij��Һ����ɫ���������գ�����ʻ�ɫ��֤�����в���K+ |
���ڳ�����0.1mol/L��ˮ��pH=11.12��������˵������ȷ���ǣ�������
| A����ˮϡ�����У�c��H+����c��OH-�� ��ֵ���� | ||
| B������������Ϣ�����ƶ�NH3?H2OΪ������� | ||
C����pH=11.12�� NaOH��Һ��ȣ�NaOH��Һ��c��Na+�� ���ڰ�ˮ��c��NH
| ||
| D����������NH4Cl ���壬��Һ��ˮ�ĵ���ƽ�⣺H2O?H++OH-�����ƶ� |
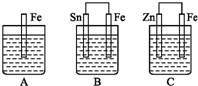 A����B��C�����ձ��зֱ�ʢ����ͬ���ʵ���Ũ�ȵ�ϡ���ᣬ��ͼ��ʾ��
A����B��C�����ձ��зֱ�ʢ����ͬ���ʵ���Ũ�ȵ�ϡ���ᣬ��ͼ��ʾ��