��Ŀ����
4��ʵ������NH4HCO3��NaHSO3�ĸ����״����ij��ȤС��Ϊ�ⶨ����NH4HCO3�ĺ�������������ڻ�����м����ʵ�鷽�����ⶨ��������������ѧС����Ҫѡ��������ҩƷ����������ʵ�飨����̨�ȹ̶�����δ���������������������ҵķ�����������˳����a��i��h��d��e��f��g��b��c���ش��������⣺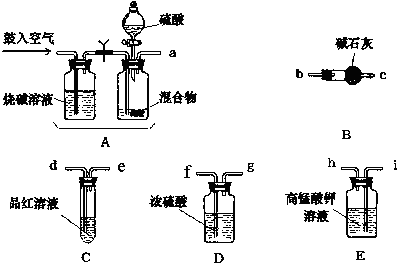
��1��A�м���������Һ�����������ǣ���Һ©����
��2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ����������ų�װ����ԭ�еĿ����������Aװ���ٴι�������������ǽ����ɵ�����ȫ���������װ�ã�
��3��Eװ�õ����������ն����������壮
��4��ʵ���У�Ҫȷ��ʵ����Cƿ��Һ����ɫ����Cƿ����Һ��ɫ����ⶨ������ܻ�ƫ�ߣ���ߡ��͡�����
��5�����������Ʒ����Ϊ13.1g��������ú��ʯ������4.4g����������NH4HCO3����������Ϊ60.3%��
��6���Ӷ����ⶨ��ȷ�Կ��ǣ�����װ�û�Ӧ����һ���ĸĽ�����B֮���ٽ�һ����ֹ������ˮ�Ͷ�����̼����B��װ�ã�
���� ����ʵ��ԭ����֪����NH4HCO3��NaHSO3�Ļ�����м������������̼�Ͷ����������壬�ø��������Һ��ȥ��������Ȼ��ͨ��Ʒ����Һ������������Ƿ���������Ž�����ͨ��Ũ����������ü�ʯ�����ն�����̼���壬��ʯ�����ӵ�������Ϊ������̼��������������ݶ�����̼�������ɼ����������NH4HCO3������ȷ��NH4HCO3�ĺ�����
��1������װ��ͼ��֪�������ƣ�
��2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ�������Ŀ�����ų�ϵͳ�еĶ�����̼���壬������ٴι������������
��ʹ���ɵ�����ȫ���������װ�ã�
��3�����������Һ�����������ն����������壮
��4����Ʒ����Һ��ɫ��˵��������������û�г�����������ʯ�����գ����Լ�ʯ��������ƫ���ٸ���ʵ��ԭ��������
��5����ʯ������4.4g��������̼���ʵ���Ϊ0.1mol����̼Ԫ���غ㣬̼��������ʵ���ҲΪ0.1mol��̼���������Ϊ7.9g��������NH4HCO3������������
��6����ʯ�������տ����еĶ�����̼��ˮ������
��� �⣺��1������װ��ͼ��֪A�м���������Һ�����������Ƿ�Һ©�����ʴ�Ϊ����Һ©����
��2��ʵ�鿪ʼ����������ǰ��Aװ����Ҫ�������Ŀ�����ų�ϵͳ�еĶ�����̼���壬������ٴι��������������ʹ���ɵ�����ȫ���������װ�ã��ʴ�Ϊ�������ɵ�����ȫ���������װ�ã�
��3�����������Һ�����������ն����������壬�ʴ�Ϊ�����ն����������壻
��4����Ʒ����Һ��ɫ��˵��������������û�г�����������ʯ�����գ����Լ�ʯ��������ƫ����ʵ��ԭ��������֪������̼���������ƫ����������NH4HCO3ƫ�ⶨ�����ƫ�ߣ��ʴ�Ϊ���ߣ�
��5����ʯ������4.4g����������̼���ʵ���Ϊ0.1mol����̼Ԫ���غ㣬̼��������ʵ���ҲΪ0.1mol��̼���������Ϊ7.9g��NH4HCO3����������Ϊ$\frac{7.9g}{13.1g}$��100%=60.3%���ʴ�Ϊ��60.3%��
��6����ʯ���������ͨ����ʯ�������տ����еĶ�����̼��ˮ����������Ӧ�ڼ�ʯ�Һ���������һ��װ�м�ʯ�ҵ�װ�ã�
�ʴ�Ϊ����B֮���ٽ�һ����ֹ������ˮ�Ͷ�����̼����B��װ�ã�
���� ������ҪĿ���Dzⶨ������̼���������������������Ҳ�ܱ���ʯ�����գ����������ն�����̼����ǰӦ���������������������ȥ������������һ�������Ը��������Һ����ˮ������Ʒ����Һ����������������Ƿ������

| A�� | ͬ��Ԫ�أ����ź�����Ӳ��������ӣ�I1������ | |
| B�� | ͨ������£�����ͬһ��Ԫ�ص�ԭ�ӣ��������I1��I2��I3 | |
| C�� | ͬ����Ԫ�أ�����仯���������ź˵���������ӣ�I1���� | |
| D�� | ͨ������£�������ԽС��Ԫ�صĽ�����Խǿ |
| A�� | ���� | B�� | ���� | C�� | ��С | D�� | ���ж� |
| A�� | �����¶ȣ�����Ӧ���ʼ�С������Ӧ�������� | |
| B�� | �����¶������������淴Ӧ�������Ӷ����̴ﵽƽ���ʱ�� | |
| C�� | �ﵽƽ��������¶Ȼ�����ѹǿ�������ڸ÷�Ӧƽ�������ƶ� | |
| D�� | �ﵽƽ������¶Ȼ��Сѹǿ�������ڸ÷�Ӧƽ�������ƶ� |
| A�� | 2H2O$\frac{\underline{\;ͨ��\;}}{\;}$ 2H2��+O2 | B�� | 2Na+2H2O�T2NaOH+H2�� | ||
| C�� | Cl2+H2O�THC1+HC1O | D�� | 2F2+2H2O�T4HF+O2 |
 ij��ѧ�С�����ʵ�飨װ����ͼ����֤�ȡ��塢�⼰�仯������й����ʣ���Ӳ�ʲ������е�A��B��C�������η���ʪ�����ɫʯ����ֽ������NaBr��Һ�������е���-KI��Һ��������ͼ��ʾ���������ͨ�������������ش��������⣺
ij��ѧ�С�����ʵ�飨װ����ͼ����֤�ȡ��塢�⼰�仯������й����ʣ���Ӳ�ʲ������е�A��B��C�������η���ʪ�����ɫʯ����ֽ������NaBr��Һ�������е���-KI��Һ��������ͼ��ʾ���������ͨ�������������ش��������⣺