��Ŀ����
 �й����ʴ�����ͼ��ʾ��ת����ϵ�����ֲ�����ʡ�ԣ���ͨ��CΪ���嵥�ʣ�GΪ�Ϻ�ɫ���嵥�ʣ�ʵ�����У����ù���E��B�Ĵ��¼�����ȡ���嵥��H��
�й����ʴ�����ͼ��ʾ��ת����ϵ�����ֲ�����ʡ�ԣ���ͨ��CΪ���嵥�ʣ�GΪ�Ϻ�ɫ���嵥�ʣ�ʵ�����У����ù���E��B�Ĵ��¼�����ȡ���嵥��H����1��C�ķ���ʽ��
��2����Ӧ�ٵĻ�ѧ����ʽΪ
��3����Ӧ�ڵ����ӷ���ʽΪ
��4��д������һ��ʵ������ȡH�Ļ�ѧ����ʽ��
��5��D��Һ��Pb��NO3��2��Һ��Ͽ��γɳ������˳�����Ksp=7.0��10-9�����������D��Һ��Pb��NO3��2��Һ��ϣ���D��Ũ��Ϊ1��10-2 mol/L�������ɳ�������Pb��NO3��2��Һ����СŨ��Ϊ
���㣺������ƶ�
ר�⣺�ƶ���
������GΪ�Ϻ�ɫ���嵥�ʣ���GΪI 2��CΪ���嵥�ʣ���C���������ԣ�D�к���I-��C��ŨA��Һ��B�ڼ��������ɣ���B����ȡ����H�����������Ƴ�BӦΪMnO 2����AΪHCl��CΪCl 2��ʵ��������MnO2����ȡ��������O 2����HΪO 2��EΪ���壬��E��KClO 3������FΪKCl��������������KClO 3���Խ�I-����ΪI 2��
Ҫ��ȷ���������ʵ�����Ʒ�����������Ʒ����������ʼȿ����������п�������Ӧ�����д������ԭ��Ӧ�����ӷ���ʽʱ����ƽ˳��Ϊ����ʧ�����غ㡢����غ㡢ԭ���غ㣮
Ҫ��ȷ���������ʵ�����Ʒ�����������Ʒ����������ʼȿ����������п�������Ӧ�����д������ԭ��Ӧ�����ӷ���ʽʱ����ƽ˳��Ϊ����ʧ�����غ㡢����غ㡢ԭ���غ㣮
���
�⣺GΪ�Ϻ�ɫ���嵥�ʣ���GΪI 2��CΪ���嵥�ʣ���C���������ԣ�D�к���I-��C��ŨA��Һ��B�ڼ��������ɣ���B����ȡ����H�����������Ƴ�BӦΪMnO 2����AΪHCl��CΪCl 2��ʵ��������MnO2����ȡ��������O 2����HΪO 2��EΪ���壬
��E��KClO3������FΪKCl��������������KClO 3���Խ�I-����ΪI2��
��1��CΪCl2���ʴ�Ϊ��Cl2��
��2����ΪMnO2��Ũ�����ڼ��������·�Ӧ�Ʊ���������Ӧ�Ļ�ѧ����ʽΪMnO2+4HCl��Ũ��
Cl2��+MnCl2+2H2O��
�ʴ�Ϊ��MnO2+4HCl��Ũ��
Cl2��+MnCl2+2H2O��
��3��ʵ��������MnO2����ȡ��������O 2����HΪO 2��EΪ���壬��E��KClO 3������FΪKCl��������������KClO 3���Խ�I-����ΪI 2����Ӧ����H+�μӣ������ԭ������ˮ����Ӧ�����ӷ���ʽΪ6I-+ClO-3+6H+=3I2+Cl-+3H2O��
�ʴ�Ϊ��6I-+ClO-3+6H+=3I2+Cl-+3H2O��
��4��ʵ������ȡ�����������ù�������ֽ������ؼ��ȷֽ⣬��Ӧ�ķ���ʽΪ2H2O2
2H2O+O2����2KClO3
2KCl+O2����
�ʴ�Ϊ��2H2O2
2H2O+O2����2KClO3
2KCl+O2����
��5���������⣬��������PbI2����Ϻ�c��I-���T5��10-3mol/L������Ksp=c[Pb��NO3��2]?c2��I-���������ɳ���ʱ�������Һ�е�Pb��NO3��2��Һ����СŨ��Ϊ
=2.8��10-4 ��mol/L�������ǰ����ԭPb��NO3��2��Һ����СŨ��Ϊ2��2.8��10-4mol/L=5.6��10-4mol/L��
�ʴ�Ϊ��5.6��10-4mol/L��
��E��KClO3������FΪKCl��������������KClO 3���Խ�I-����ΪI2��
��1��CΪCl2���ʴ�Ϊ��Cl2��
��2����ΪMnO2��Ũ�����ڼ��������·�Ӧ�Ʊ���������Ӧ�Ļ�ѧ����ʽΪMnO2+4HCl��Ũ��
| ||
�ʴ�Ϊ��MnO2+4HCl��Ũ��
| ||
��3��ʵ��������MnO2����ȡ��������O 2����HΪO 2��EΪ���壬��E��KClO 3������FΪKCl��������������KClO 3���Խ�I-����ΪI 2����Ӧ����H+�μӣ������ԭ������ˮ����Ӧ�����ӷ���ʽΪ6I-+ClO-3+6H+=3I2+Cl-+3H2O��
�ʴ�Ϊ��6I-+ClO-3+6H+=3I2+Cl-+3H2O��
��4��ʵ������ȡ�����������ù�������ֽ������ؼ��ȷֽ⣬��Ӧ�ķ���ʽΪ2H2O2
| ||
| �� |
| ||
| �� |
�ʴ�Ϊ��2H2O2
| ||
| �� |
| ||
| �� |
��5���������⣬��������PbI2����Ϻ�c��I-���T5��10-3mol/L������Ksp=c[Pb��NO3��2]?c2��I-���������ɳ���ʱ�������Һ�е�Pb��NO3��2��Һ����СŨ��Ϊ
| 7.0��10-9 |
| (5��10-3)2 |
�ʴ�Ϊ��5.6��10-4mol/L��
���������⿼��Ԫ�ػ�������ƶϣ���Ŀ�Ѷ��еȣ������漰������ԭ��Ӧԭ�����ܶȻ��ȵļ������һ�����ۺ��ԣ�ע��������ʵ�������Ϊ���ƶ����ͻ�ƿڣ�

��ϰ��ϵ�д�
 �����Ƹ���ʦ����ϵ�д�
�����Ƹ���ʦ����ϵ�д� ��ͨ����ͬ����ϰ��ϵ�д�
��ͨ����ͬ����ϰ��ϵ�д� ����С����ͬ������ϵ�д�
����С����ͬ������ϵ�д�
�����Ŀ
��ʵ��I-IV��ʵ������Ԥ�������ǣ�������
A��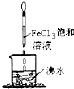 ���������Һ�����ɫ��ֹͣ���ȣ�������ͨ����ϵʱ�ɲ��������ЧӦ |
B��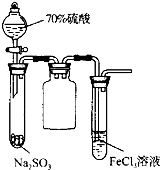 ʵ���Թ��г��ֵ���ɫ���� |
C��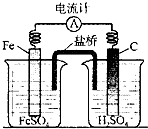 ʵ����̼��������ɫ���ݲ��� |
D��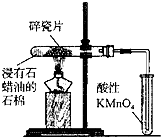 ʵ��������KMnO4��Һ�г�����������ɫ����ȥ |
Ԫ�����ڱ���Ԫ�������ɿ���ָ�����ǽ��й����Ե��Ʋ���жϣ�����˵���������ǣ�������
| A����X��Y������̬�⻯�����Է���������X��Y����е㣺X��Y |
| B����R2-��M+�ĵ��Ӳ�ṹ��ͬ����ԭ��������R��M |
| C����X��Y����ͬ����Ԫ�أ������ԭ��������X��Y����Xʧ����������Yǿ |
| D����M��N��ͬ����Ԫ�أ���ԭ��������M��N����ǽ����ԣ�M��N |
�����й����ʵ����ʻ�Ӧ�õ�˵��������ǣ�������
| A��������Ũ���������������ۻ������ڳ�����������������������Ũ���� |
| B���������ᴿ�����ʲ��������������� |
| C���������費���κ��ᷴӦ������ʯӢ������������ |
| D��п�Ľ��������Ա�����ǿ�����ں��������װ����п���Լ����丯ʴ |
����ʵ���������ȷ���ǣ�������
| A����������ʱ��Ӧʹ������е�ˮ����ȫ���ɺ���ֹͣ���� |
| B���������ʱ��Ӧʹ�¶ȼ�ˮ�������Һ������ |
| C����Һ����ʱ����Һ©�����²�Һ����¿ڷų����ϲ�Һ����Ͽڵ��� |
| D����ȡ����ʱ��Ӧѡ���л���ȡ��������ȡ�����ܶȱ����ˮ�� |
�����й��л����˵����ȷ���ǣ�������
| A�������ʡ���ά�ء����ǡ���Ȼ�����۶��Ǹ߷��ӻ����� |
| B����ϩ�ͱ�����ʹ��ˮ��ɫ����ɫ��ԭ����ͬ |
| C�����ۡ������ǡ�֬���͵�������һ�������¶��ܷ���ˮ�ⷴӦ |
| D���Ʊ���Ĺ����漰��������Ӧ |
�������ʵ�ˮ��Һ�ܵ��磬�����ڷǵ���ʵ��ǣ�������
| A��HNO3 |
| B��C2H5OH |
| C��NH4NO3 |
| D��CO2 |
 A��B��C�Ƕ����ڷǽ���Ԫ�أ��˵������������Aԭ����Χ�����Ų�Ϊns2np2��C�ǵؿ��к�������Ԫ�أ�D��E�ǵ�������Ԫ�أ�����EԪ�صĺ˵����Ϊ29��Dԭ�Ӻ���δ�ɶԵ�������ͬ��������࣮���ö�Ӧ��Ԫ�ط��Ż�ѧʽ��գ�
A��B��C�Ƕ����ڷǽ���Ԫ�أ��˵������������Aԭ����Χ�����Ų�Ϊns2np2��C�ǵؿ��к�������Ԫ�أ�D��E�ǵ�������Ԫ�أ�����EԪ�صĺ˵����Ϊ29��Dԭ�Ӻ���δ�ɶԵ�������ͬ��������࣮���ö�Ӧ��Ԫ�ط��Ż�ѧʽ��գ�