��Ŀ����
�л���A�Ľṹ��ʽΪ��

��1��A�ķ���ʽΪ ��
��2��A��NaOHˮ��Һ�м��ȣ����ữ�õ��л���B��D��D�Ƿ����廯�������1molA������Ӧʱ��������� mol NaOH��
��3��B��һ�������·���������Ӧ������ij��Ԫ����������Ԫ�����Ľṹ��ʽΪ ��
��4��д��������ֻ��һ��ȡ���������������D������ͬ���칹��Ľṹ��ʽ ��
��5��D��ijͬϵ���������г��ȼ�����ɶ�����̼��ˮ������n��CO2����n��H2O��=3��2����ͬϵ��ķ���ʽΪ ��

��1��A�ķ���ʽΪ
��2��A��NaOHˮ��Һ�м��ȣ����ữ�õ��л���B��D��D�Ƿ����廯�������1molA������Ӧʱ���������
��3��B��һ�������·���������Ӧ������ij��Ԫ����������Ԫ�����Ľṹ��ʽΪ
��4��д��������ֻ��һ��ȡ���������������D������ͬ���칹��Ľṹ��ʽ
��5��D��ijͬϵ���������г��ȼ�����ɶ�����̼��ˮ������n��CO2����n��H2O��=3��2����ͬϵ��ķ���ʽΪ
���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������1���ɽṹ��ʽȷ���л��ﺬ�е�Ԫ�������ԭ����Ŀ����ȷ���л������ʽ��
��2��A��NaOHˮ��Һ�м��ȣ����ữ�õ��л���B��D��D�Ƿ����廯�����BΪ ��DΪ
��DΪ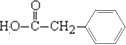 ��
��
��3��BΪ �������Ȼ����ǻ����ɷ���������Ӧ��
�������Ȼ����ǻ����ɷ���������Ӧ��
��4���γɵ�����Ϊ��������������ᱽ�����Լ����ᱽ�״�����
��5��DΪ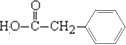 ����Ӧͬϵ���ͨʽΪCnH��2n-8��O2�����n��CO2����n��H2O��=3��2���㣮
����Ӧͬϵ���ͨʽΪCnH��2n-8��O2�����n��CO2����n��H2O��=3��2���㣮
��2��A��NaOHˮ��Һ�м��ȣ����ữ�õ��л���B��D��D�Ƿ����廯�����BΪ
 ��DΪ
��DΪ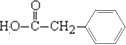 ��
����3��BΪ
 �������Ȼ����ǻ����ɷ���������Ӧ��
�������Ȼ����ǻ����ɷ���������Ӧ����4���γɵ�����Ϊ��������������ᱽ�����Լ����ᱽ�״�����
��5��DΪ
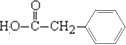 ����Ӧͬϵ���ͨʽΪCnH��2n-8��O2�����n��CO2����n��H2O��=3��2���㣮
����Ӧͬϵ���ͨʽΪCnH��2n-8��O2�����n��CO2����n��H2O��=3��2���㣮���
�⣺��1���ɽṹ��ʽ��֪A�ķ���ʽΪC12H13O4Cl���ʴ�Ϊ��C12H13O4Cl��
��2��A��NaOHˮ��Һ�м��ȣ����ữ�õ��л���B��D��D�Ƿ����廯�����BΪ ��DΪ
��DΪ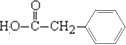 ��
��
A�к����Ȼ��������������Ʒ����кͷ�Ӧ������������Clԭ�ӣ�ˮ�������Ȼ���HCl����1molA������Ӧʱ���������3mol NaOH��
�ʴ�Ϊ��3��
��3��BΪ �������Ȼ����ǻ����ɷ���������Ӧ���γ���Ԫ���Ľṹ��ʽΪ
�������Ȼ����ǻ����ɷ���������Ӧ���γ���Ԫ���Ľṹ��ʽΪ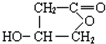 ��
��
�ʴ�Ϊ��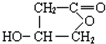 ��
��
��4��DΪ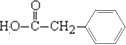 ����Ӧ��ͬ���칹���У��γɵ�����Ϊ��������������ᱽ�����Լ����ᱽ�״������ṹ��ʽ�ֱ�Ϊ
����Ӧ��ͬ���칹���У��γɵ�����Ϊ��������������ᱽ�����Լ����ᱽ�״������ṹ��ʽ�ֱ�Ϊ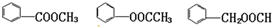 ��
��
�ʴ�Ϊ��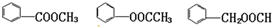 ��
��
��5��DΪ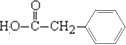 ����Ӧͬϵ���ͨʽΪCnH��2n-8��O2��n��CO2����n��H2O��=3��2����n����2n-8��=3��4��n=12�������ʽΪC12H16O2��
����Ӧͬϵ���ͨʽΪCnH��2n-8��O2��n��CO2����n��H2O��=3��2����n����2n-8��=3��4��n=12�������ʽΪC12H16O2��
�ʴ�Ϊ��C12H16O2��
��2��A��NaOHˮ��Һ�м��ȣ����ữ�õ��л���B��D��D�Ƿ����廯�����BΪ
 ��DΪ
��DΪ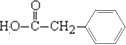 ��
��A�к����Ȼ��������������Ʒ����кͷ�Ӧ������������Clԭ�ӣ�ˮ�������Ȼ���HCl����1molA������Ӧʱ���������3mol NaOH��
�ʴ�Ϊ��3��
��3��BΪ
 �������Ȼ����ǻ����ɷ���������Ӧ���γ���Ԫ���Ľṹ��ʽΪ
�������Ȼ����ǻ����ɷ���������Ӧ���γ���Ԫ���Ľṹ��ʽΪ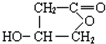 ��
���ʴ�Ϊ��
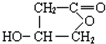 ��
����4��DΪ
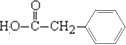 ����Ӧ��ͬ���칹���У��γɵ�����Ϊ��������������ᱽ�����Լ����ᱽ�״������ṹ��ʽ�ֱ�Ϊ
����Ӧ��ͬ���칹���У��γɵ�����Ϊ��������������ᱽ�����Լ����ᱽ�״������ṹ��ʽ�ֱ�Ϊ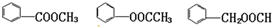 ��
���ʴ�Ϊ��
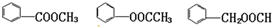 ��
�� ��5��DΪ
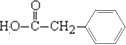 ����Ӧͬϵ���ͨʽΪCnH��2n-8��O2��n��CO2����n��H2O��=3��2����n����2n-8��=3��4��n=12�������ʽΪC12H16O2��
����Ӧͬϵ���ͨʽΪCnH��2n-8��O2��n��CO2����n��H2O��=3��2����n����2n-8��=3��4��n=12�������ʽΪC12H16O2���ʴ�Ϊ��C12H16O2��
���������⿼���л�����ƶϣ�Ϊ��Ƶ���㣬��Ŀ�ѶȲ���ע������л�������ŵ����ʣ�Ϊ��������Ŀ�Ĺؼ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
�����£����и���������ָ����Һ��һ���ܴ���������ǣ�������
| A����Al��Ӧ�ų�H2����Һ��Mg2+��Ca2+��HCO3-��NO3- | ||
| B��10mol?L-1��Ũ��ˮ��Al3+��NH4+��NO3-��I- | ||
| C��0.1 mol?L-1KMnO4��Һ��Na+��Fe2+��SO42-��Cl- | ||
D��
|
���й��ڵ���ƽ�ⳣ����K����˵������ȷ���ǣ�������
| A������ƽ�ⳣ����K��ԽС����ʾ������ʵ�������Խ�� |
| B������ƽ�ⳣ����K�����¶��� |
| C����ͬŨ�ȵ�ͬһ������ʣ������ƽ�ⳣ����K����ͬ |
| D����Ԫ�����������ƽ�ⳣ�����ϵΪ��K1��K2��K3 |
ʵ���ǻ�ѧ�о��Ļ�����ͼ����ʾ��ʵ�鷽����װ�û������ȫ��ȷ���ǣ�������
��֪�����������ڱ���ʳ��ˮ��
��֪�����������ڱ���ʳ��ˮ��
A�� ��ȥ���е��������� |
B�� Ũ����ϡ�� |
C�� �����AgNO3��Һ��AgCl |
D�� ����30.46g��NaOH���� |
 ��ͼ��ijѧУʵ���Ҵӻ�ѧ�Լ��̵�����������������Լ���ǩ�ϵIJ������ݣ�ijͬѧ�Ӹ��Լ�ƿ��ȡ��20.0g���壬���1L��Һ������㣺
��ͼ��ijѧУʵ���Ҵӻ�ѧ�Լ��̵�����������������Լ���ǩ�ϵIJ������ݣ�ijͬѧ�Ӹ��Լ�ƿ��ȡ��20.0g���壬���1L��Һ������㣺 �ϳɰ���ҵ�ĺ��ķ�Ӧ��N2��g��+3H2��g��
�ϳɰ���ҵ�ĺ��ķ�Ӧ��N2��g��+3H2��g�� ��֪ˮ��25���95��ʱ�������ƽ��������ͼ��ʾ��
��֪ˮ��25���95��ʱ�������ƽ��������ͼ��ʾ��