��Ŀ����
7��A��B���ֽ��������ʵ�����Ϊa molʱ���ֱ������������ᷴӦ����������������2ag��A��B��һ�������£��ֱ�������ʵ�����������Ӧ������A��B�����ʵ���֮��Ϊ3��2�������ʵ�����A��B��ɵĻ����19.2g�����������ᷴӦ�������������ڱ�״���µ����Ϊ8.96L����������B��A��ԭ�Ӹ�����Ϊ1��1.4����1���Է������ǵĻ��ϼۣ�
��2����A��B�����ԭ��������
��3����ȷ��A��B����ʲô������
��4��A��B���γɵľ����У������������ǿ����Fe��������һ�жϵ������������۵�ȸƸߣ��������Ľ�����û�иƻ��ã��ʽ�����ǿ��
���� ��1�����ݻ��ϼ������غ�ȷ��A��B�Ļ��ϼۣ�
��2��ͨ������ʽA+2HCl=ACl2+H2����B+2HCl=BCl2+H2��������A��B�����ԭ�������ֱ�Ϊx��y����������ɽ������ʽ��ϵ����x��y���ɣ�
��3�������ԭ������ȷ��Ԫ�أ�
��4�������۵㣬���������жϣ�
��� �⣺��1��n��H2��=$\frac{2ag}{2g/mol}$=amol����H+��H2���ϼ۽���2����amolH2����2amol����A��An+�����Ͻ�����n����amolA�����Ͻ�����anmol�����ݻ��ϼ������غ�ã�2a=an�����n=2��Ԫ��BҲͬ����ˣ�
��Ԫ��A��B�Ļ��ϼ۶�Ϊ+2�ۣ�
��2����n��H2��=$\frac{8.96}{22.4}$=0.4mol
��A��B�����ԭ�������ֱ�Ϊx��y������μӷ�Ӧ��A��B��Ϊamol��
A+2HCl=ACl2+H2��
1 1
a a
B+2HCl=BCl2+H2��
1 1
a a
$\left\{\begin{array}{l}{ax+ay=19.2}\\{a+a=0.4}\end{array}\right.$����a=0.2mol����0.2x+0.2y=19.2�٣�����Ϊ��������B��A��ԭ�Ӹ�����Ϊ1��1.4����$\frac{\frac{m}{x}}{\frac{m}{y}}=\frac{1}{1.4}$����$\frac{y}{x}=\frac{5}{7}$�ڣ����ڴ���ٵã�x=40��y=56����A�����ԭ������Ϊ40��B�����ԭ������Ϊ56��
��A�����ԭ������Ϊ40��B�����ԭ������Ϊ56��
��3��A�����ԭ������Ϊ40����ΪCa��B�����ԭ������Ϊ56����ΪFe��
��Ԫ��AΪCa��Ԫ��BΪFe��
��4�������۵�ȸƸߣ��������Ľ�����û�иƻ��ã��ʽ�����ǿ���ʴ�Ϊ��Fe�������۵�ȸƸߣ��������Ľ�����û�иƻ��ã��ʽ�����ǿ��
���� �����ѶȽϴ���Ҫ�ǻ����û��ϼ������غ㣬ȥ����Ԫ�صĻ��ϼۣ��Լ��۷е㣬���������ԶԽ�������Ӱ�죮

 53���ò�ϵ�д�
53���ò�ϵ�д�| A�� | �ܱ�������9.6�������11.2�����ۻ�ϼ�������������17.6��ʱ�ų�19.12 kJ��������Fe��S��+S��S��=FeS��S����H=-95.6 kJ•mol-1 | |
| B�� | ϡ�����0.1mol/L������������Һ��Ӧ��H+��aq��+OH-��aq��=H2O��l����H=-57.3kJ/mol | |
| C�� | ��֪1 mol������ȫȼ������Һ̬ˮ���ų�������Ϊ285.5 kJ����ˮ�ֽ���Ȼ�ѧ����ʽ��2H2O��l��=2H2��g��+O2��g����H=+285.5kJ/mol | |
| D�� | ��֪2C��s��+O2��g��=2CO��g����H=-221 kJ/mol�����֪C��ȼ���ȡ�H=-110.5 kJ/mol |
��
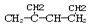 ������1��3-����-2-��ϩ
������1��3-����-2-��ϩ����Է���������ͬ�������л���һ����ͬ���칹��
��ϩ����ͬϵ����̼���������������
�ܴ�ú�����з���������ױ������ױ����л���Ĺ���Ϊ��ѧ�仯
������ͬ�����£�ȼ�յ�������C2H4�ͱ�����ϩ����O2������
�ޱ���Ȳ�������6��ԭ����ͬһ��ֱ���ϣ�
| A�� | ������ȷ | B�� | �ۢݢ���ȷ | C�� | �ڢܢ���ȷ | D�� | �٢ۢ���ȷ |
| A�� | SO2+Br2+H2O�TH2SO4+2HBr | B�� | SO2+H2O�TH2SO3 | ||
| C�� | SO2+2NaOH�TNa2SO3+H2O | D�� | SO2+CaO$\frac{\underline{\;����\;}}{\;}$CaSO3 |
| ������Һ/mL | 20.0 | 40.0 | 60.0 | 80.0 |
| ʣ�����/g | 9.36 | 7.12 | 4.88 | 3.20 |
��1����Ʒ������ͭ�����ʵ���֮��n��Fe����n��Cu��=3��1��
��2���ڷ�Ӧ�����ϵ�У��ټ���4.0mol/L��H2O2��Һ25.0mL������������������Һ����ַ�Ӧ����Һ�д��ڵĸ��������ӵ����ʵ����ֱ��Ƕ��٣�
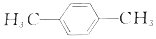 ����Ա�����ȩ��
����Ա�����ȩ��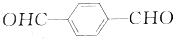 �����ɱ�����ֱ������Ϊ�Ա������ᣬǰ�����ַ����У��Ʊ������ĶԱ�������ʱ�����������ĵ��������ʵ���֮��Ϊ��������
�����ɱ�����ֱ������Ϊ�Ա������ᣬǰ�����ַ����У��Ʊ������ĶԱ�������ʱ�����������ĵ��������ʵ���֮��Ϊ��������| A�� | 3��1 | B�� | 2��1 | C�� | 1��1 | D�� | 4��1 |
| A�� | пΪ���� | B�� | ���Ӵ�п������ | ||
| C�� | п������������Ӧ | D�� | ���Һ�϶�����ǿ�� |
| A�� | 0.1 mol/L ���������Һ�У�c��NH4+����c��SO42?����c��H+����c��OH?�� | |
| B�� | 0.2mol/LNaHCO3��Һ��0.1mol/LNaOH��Һ�������ϣ�c��H+���Tc��OH?��+c��HCO3?��+2c��H2CO3�� | |
| C�� | pH֮��Ϊ14��H2C2O4��KOH ��Һ��ϣ�c��K+����c��H+����c��OH?����c��HC2O4?�� | |
| D�� | pH=5 ��H2S��Һ�У�c��H+����c��HS?��=1��10-5mol/L |