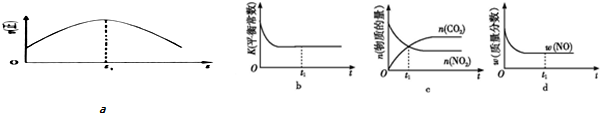
��Ŀ����
4��A��Q10���л����ת����ϵ��ͼ��ʾ�����в��ַ�Ӧ������δ��������֪��G�����������ܷ���������Ӧ�������ʵ�����A��J�ֱ���������NaHCO3��Һ��Ӧ������CO2����ͬ״�����������Ϊ1��2��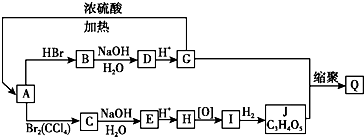
��1��A�Ľṹ��ʽΪCH2=CHCOOH��H�Ľṹ��ʽΪCH2OHCHOHCOOH�� J�Ľṹ��ʽΪHOOCCHOHCOOH��
��2��д�����л�ѧ��Ӧ�ķ�Ӧ����
A��C�ӳɷ�Ӧ��C��Eȡ����Ӧ��
��3��д������ת���Ļ�ѧ����ʽ
B��DBrCH2CH2COOH+2NaOH $��_{��}^{H_{2}O}$HOCH2CH2COONa+NaBr+H2O��G��ACH2OHCH2COOH $��_{��}^{Ũ����}$CH2=CHCOOH+H2O��
��4����G������ͬ�����ŵ����ʵĽṹ��ʽ����CH3CHOHCOOH�� G����һ��ͬ���칹��ɷ���������Ӧ����˴Ź�����������5���壬��ṹ��ʽΪHOCH2CHOHCHO��
��5��Q�Ľṹ��ʽ
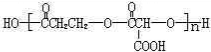 ��
��
���� A�������HBr�����ӳɷ�Ӧ��˵������C=C��������NaHCO3��Һ��Ӧ��˵������-COOH����J�ķ���ʽ��֪A����3��Cԭ�ӣ���AΪCH2=CHCOOH��G�����������������Ӧ��˵������ȩ������GӦΪHOCH2CH2COOH��DΪHOCH2CH2COONa��BΪBrCH2CH2COOH��CΪCH2BrCHBrCOOH��EΪCH2OHCHOHCOONa��HΪCH2OHCHOHCOOH��IΪHOOCCOCOOH��G��J�ܷ������۷�Ӧ������Ӧ����-OH��-COOH����J����5��Oԭ�ӣ���J��Ӧ��2��-COOH��G����2��-OH������J�ĽṹΪ��JΪHOOCCHOHCOOH��QΪ ����϶�Ӧ�л���Ľṹ�������Լ���ĿҪ��ɽ����⣮
����϶�Ӧ�л���Ľṹ�������Լ���ĿҪ��ɽ����⣮
��� �⣺A�������HBr�����ӳɷ�Ӧ��˵������C=C��������NaHCO3��Һ��Ӧ��˵������-COOH����J�ķ���ʽ��֪A����3��Cԭ�ӣ���AΪCH2=CHCOOH��G�����������������Ӧ��˵������ȩ������GӦΪHOCH2CH2COOH��DΪHOCH2CH2COONa��BΪBrCH2CH2COOH��CΪCH2BrCHBrCOOH��EΪCH2OHCHOHCOONa��HΪCH2OHCHOHCOOH��
IΪHOOCCOCOOH��JΪHOOCCHOHCOOH��QΪ ��
��
��1�������Ϸ�����֪AΪCH2=CHCOOH��HΪCH2OHCHOHCOOH��JΪHOOCCHOHCOOH���ʴ�Ϊ��CH2=CHCOOH��CH2OHCHOHCOOH��HOOCCHOHCOOH��
��2��CΪBrCH2CHBrCOOH��A�����ӳɷ�Ӧ����C��EΪCH2OHCHOHCOONa��C����ȡ����Ӧ����E��
�ʴ�Ϊ���ӳɣ�ȡ����
��3��BΪBrCH2CH2COOH��DΪHOCH2CH2COONa��B������������Һ�з���ˮ�ⷴӦ����D����Ӧ�ķ���ʽΪBrCH2CH2COOH+2NaOH$��_{��}^{H_{2}O}$HOCH2CH2COONa+NaBr+H2O��G��A�ķ�Ӧ��HOCH2CH2COOH��Ũ����������¼��ȷ�����ȥ��Ӧ����CH2=CHCOOH����Ӧ�Ļ�ѧ����ʽΪ��CH2OHCH2COOH $��_{��}^{Ũ����}$CH2=CHCOOH+H2O��
�ʴ�Ϊ��BrCH2CH2COOH+2NaOH $��_{��}^{H_{2}O}$HOCH2CH2COONa+NaBr+H2O��CH2OHCH2COOH $��_{��}^{Ũ����}$CH2=CHCOOH+H2O��
��4����G������ͬ�����ŵ��������ǻ�λ���칹�����ʵĽṹ��ʽΪ��CH3CHOHCOOH��GΪHOCH2CH2COOH����һ��ͬ���칹��ɷ���������Ӧ��˵������ȩ������˴Ź�����������5���壬GΪHO-CH2-CH2-COOH������ͬ�������к���-CHOʱ˵���ǽ�ԭ����-COOH��Ϊ-CHO��-OH����ṹ��ʽΪHOCH2CHOHCHO��
�ʴ�Ϊ��CH3CHOHCOOH��HOCH2CHOHCHO��
��5�������Ϸ�����֪QΪ ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
���� ���⿼���л�����ƶϣ�Ϊ�߿��������ͣ�������ѧ���ķ����������ۺϰ����л���Ľṹ�����ʵ��������Ѷ��еȣ�ע����AΪ�������ͻ�ƿڣ�

| A�� | ��������ͬ������һ������ͬһ��Ԫ�� | |
| B�� | ͬλ�ص����ʼ�����ȫ��ͬ | |
| C�� | ��������ͬ��������Ҳ��ͬ���������ӣ���������һ�ַ��Ӻ�һ������ | |
| D�� | ij��Ԫ�ص����ԭ������ȡ������������������ |
�ټ�������Һ��Ũ����--��ɫ �ڵ�����Һ��������--��ɫ
���廯������ֽ�--��ɫ ���ȵ�����ͭ���Ҵ�--��ɫ
��������ˮ���ú�--��ɫ ���������ڿ����о���--����ɫ
��Fe3+����������--��ɫ ��ʯ����Һ��ͨ������������--��ɫ��������
| A�� | �٢ڢۢ� | B�� | �٢ܢݢ� | C�� | �ڢۢܢ� | D�� | �ܢݢߢ� |
| A�� | N2�����壩+O2�����壩?2NO�����壩 ����Ӧ���� | |
| B�� | NH4HCO3�����壩?NH3�����壩+H2O�����壩+CO2�����壩 ����Ӧ���� | |
| C�� | 3O2�����壩?2O3�����壩 ����Ӧ���� | |
| D�� | 2NO2�����壩?N2O4�����壩 ����Ӧ���� |
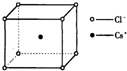 ��ͼ���Ȼ�菉���ľ�������֪������2�������Cs+���Ӻ˼��Ϊa cm���Ȼ�蘆�Ħ������ΪM�����Ȼ�菉�����ܶ�Ϊd g•cm-3�������ӵ�������ֵ�ɱ�ʾΪ��������
��ͼ���Ȼ�菉���ľ�������֪������2�������Cs+���Ӻ˼��Ϊa cm���Ȼ�蘆�Ħ������ΪM�����Ȼ�菉�����ܶ�Ϊd g•cm-3�������ӵ�������ֵ�ɱ�ʾΪ��������| A�� | 2Ma3d | B�� | M/��a3d�� | C�� | M/��2a3d�� | D�� | 2M/��a3d�� |
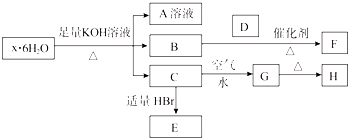
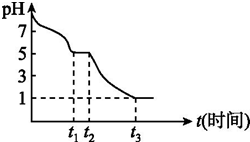 �����������ƣ�Na2S2O4���ֳƱ��շۣ���ӡˢ��ҵ����Ҫ�Ļ�ԭ����ij����С���������ʵ�飮
�����������ƣ�Na2S2O4���ֳƱ��շۣ���ӡˢ��ҵ����Ҫ�Ļ�ԭ����ij����С���������ʵ�飮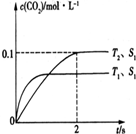 Ŀǰ����̼���š����ܹ�ע��CO2�IJ�������Ч�������ó�Ϊ��ѧ���о�����Ҫ���⣮
Ŀǰ����̼���š����ܹ�ע��CO2�IJ�������Ч�������ó�Ϊ��ѧ���о�����Ҫ���⣮