��Ŀ����
ijͬѧ����ˮ�ʼ��վ����480 mL 0��5 mol��L-1NaOH��Һ�Ա�ʹ�á�
��1����ͬѧӦѡ������mL������ƿ��
��2���������������ͼ��ʾ�����ͼ����Ӧ����ͼ�еġ�����(��ѡ����ĸ)֮�䡣
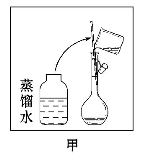
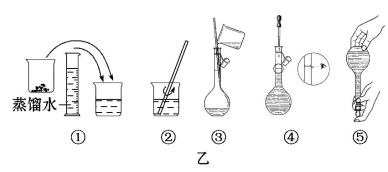
A������ۡ�������B������ڡ�������C�������
��3����ͬѧӦ��ȡNaOH���塡������g��������Ϊ23��1 g���ձ�����������ƽ�ϳ�ȡ����NaOH����ʱ�����ڸ�����ѡȡ����������С��������(����ĸ)��������ͼ��ѡ������ȷ��ʾ����λ�õ�ѡ�������(����ĸ)��
������������
| a | b | c | d | e |
�����С/g | 100 | 50 | 20 | 10 | 5 |
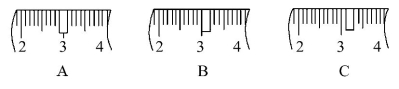
��1��500����2��C����3��10��0��c��d��C
��������
�����������1��ѡ������ƿʱ������ƿ���ݻ�Ӧ��������Һ�������ͬ�����������Ӧѡ��500 mL������ƿ��
��2����ͼ������ϴ���ձ��ڱںͲ�����������ϴ��Һת��������ƿ�У����Ӧ���ڢۺ͢�֮�䡣
��3��m(NaOH)=0��5 L��0��5 mol��L-1��40 g��mol-1=10��0 g������ʱ��Ӧ����20 g��10 g���룬���ƶ�������3��1 g��
���㣺��Һ�����Ƽ�����

�±�Ϊ�������������е����������Լ���ȥ��Щ����Ӧѡ�õ��Լ��������������ȷ��һ��Ϊ��(����)
��� | ���� | ���� | ����Ӧѡ�õ��Լ���������� |
�� | KNO3��Һ | KOH | ����FeCl3��Һ�������� |
�� | FeSO4��Һ | CuSO4 | ����������ۣ������� |
�� | H2 | CO2 | ͨ��ʢ��NaOH��Һ��ϴ��ƿ����ͨ��ʢ��Ũ�����ϴ��ƿ |
�� | NaNO3���� | CaCO3 | �ܽ⡢���� ������ |
A���٢ڢۢ� B���ۢ� C���ڢۢ� D���٢ڢ�