��Ŀ����
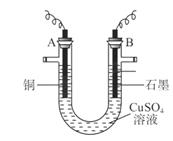
(10��)ij�о���ѧϰС���õ�� ��Һ�ķ����ⶨͭ�����ԭ������(װ����ͼ),ʵ���в��ij�缫����������Ϊm g����֪��Ԫ�����ԭ������Ϊ16����ش�:
��Һ�ķ����ⶨͭ�����ԭ������(װ����ͼ),ʵ���в��ij�缫����������Ϊm g����֪��Ԫ�����ԭ������Ϊ16����ش�: 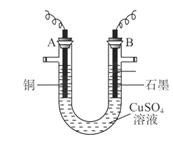
(1)m gӦΪ ������������(��A��B)������ܷ�Ӧ�����ӷ���ʽΪ ��
(2)��ʵ��(��� ��Һ)�У�ѧϰС������������ⶨͭ�����ԭ��������
��Һ)�У�ѧϰС������������ⶨͭ�����ԭ��������
�ٵ�һ�����У�ͬѧ�Dz����������ΪV L(��״��)����ͭ�����ԭ��������(��m��V��ʾ) ��
�ڵڶ������У�����������Ҫ�ⶨ����(�����) ��
a. A�����ӵ����� b. ����װ�ü��ٵ�����
c. ������Һ����� d. ������Һ��pH�����ɴ˼������Һ��H+�����ʵ���Ũ�ȣ�
(1)A 2CuSO4��2H2O 2Cu��2H2SO4��O2�� (2) ��
2Cu��2H2SO4��O2�� (2) ��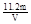 ��ab��acd
��ab��acd
����

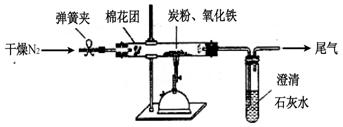
(10��)ij�о���ѧϰС��Ϊ��̽������ĵ������������������ʵ�顣
ʵ��һ ���ƴ�����Һ��Ũ��ȡ����������250 mL 0.2 mol/L�Ĵ�����Һ����0.2 mol/L�Ĵ�����Һϡ�ͳ�����Ũ�ȵ���Һ������NaOH����Һ�����������Һ��Ũ�Ƚ��б궨���ش��������⣺
����250 mL 0.2 mol/L ������Һʱ��Ҫ�õ��IJ�����������Ͳ���ձ�����������________________��_________________��
ʵ��� ̽��Ũ�ȶԴ������̶ȵ�Ӱ��
��pH�Ʋⶨ25��ʱ��ͬŨ�ȵĴ����pH���������£�
| ����Ũ��(mol/L) | 0.0010 | 0.0100 | 0.0200 | 0.1000 | 0.2000 |
| pH | 3.88 | 3.38 | 3.23 | 2.88 | 2.73 |
�Ǹ��ݱ������ݣ����Եó�������������ʵĽ��ۣ�����Ϊ�ó��˽��۵�������
_______________________________________________________________________________
_____________________________________________
 ____________________________________
____________________________________�ȴӱ��е����ݣ������Եó���һ���ۣ����Ŵ���Ũ�ȵļ�С������ĵ���̶�____________��
(��������)
ʵ���� ̽���¶ȶԴ������̶ȵ�Ӱ��
�������һ��ʵ����ɸ�̽������������ʵ�鷽��_________________________
_____________________________________________��

 ��Һ�ķ����ⶨͭ�����ԭ������(װ����ͼ),ʵ���в��ij�缫����������Ϊm g����֪��Ԫ�����ԭ������Ϊ16����ش�:
��Һ�ķ����ⶨͭ�����ԭ������(װ����ͼ),ʵ���в��ij�缫����������Ϊm g����֪��Ԫ�����ԭ������Ϊ16����ش�: