��Ŀ����
��һ��ɫ��������Һ�����ܺ��нϴ�����Cu2+��Ba2+��
��N
��I-��S2-��H+��K+�����������ֻ���֣�������ʵ�飺
��ȡ��Һ������������������ų���
����ȡԭ��Һ��Na2SO3��Һ��������ų���Ҳ�а�ɫ�������ɣ��ټ������������ȫ��ʧ��
����ȡԭ��Һ����AgNO3��Һ�������ɣ�
��1���ɴ��ж�ԭ��Һ��һ�����е������� ��һ�������е������� �����ܺ��е������� ��
��2���йط�Ӧ�ٵ����ӷ���ʽΪ ��
| SO | 2- 4 |
| O | - 3 |
��ȡ��Һ������������������ų���
����ȡԭ��Һ��Na2SO3��Һ��������ų���Ҳ�а�ɫ�������ɣ��ټ������������ȫ��ʧ��
����ȡԭ��Һ����AgNO3��Һ�������ɣ�
��1���ɴ��ж�ԭ��Һ��һ�����е�������
��2���йط�Ӧ�ٵ����ӷ���ʽΪ
���㣺���ʵļ���ͼ����ʵ�鷽�����
ר�⣺���ʼ��������
��������һ��ɫ��������Һ������ɫ����һ�������ڣ�
��ȡ��Һ������������������ų������������ӣ����ܺ���NO3-��
����ȡԭ��Һ��Na2SO3��Һ��������ų����������Ƕ�������Ҳ�а�ɫ�������ɣ��ټ������������ȫ��ʧ�����Գ��������ᱵ�����ܺ�����������ӣ����Ǻ�����������ӣ��������������Ӷ����ڣ�
����ȡԭ��Һ����AgNO3��Һ�������ɣ���ԭ����Һһ������I-��S2-��SO42-���ݴ˻ش��жϣ�
��ȡ��Һ������������������ų������������ӣ����ܺ���NO3-��
����ȡԭ��Һ��Na2SO3��Һ��������ų����������Ƕ�������Ҳ�а�ɫ�������ɣ��ټ������������ȫ��ʧ�����Գ��������ᱵ�����ܺ�����������ӣ����Ǻ�����������ӣ��������������Ӷ����ڣ�
����ȡԭ��Һ����AgNO3��Һ�������ɣ���ԭ����Һһ������I-��S2-��SO42-���ݴ˻ش��жϣ�
���
�⣺��1����һ��ɫ��������Һ������ɫ����Cu2+һ�������ڣ�
��ȡ��Һ������������������ų������������ӣ����ܺ���NO3-��
����ȡԭ��Һ��Na2SO3��Һ��������ų����������Ƕ�������Ҳ�а�ɫ�������ɣ��ټ������������ȫ��ʧ�����Գ��������ᱵ��һ�����б����ӣ�
һ����������������ӣ�һ��������������ӣ�����ȡԭ��Һ����AgNO3��Һ�������ɣ���ԭ����Һһ������I-��S2-��SO42-��
���Ͽ�֪����Һ��һ������H+��NO3-��Ba2+�����ܺ��е�������K+��һ������Cu2+��I-��S2-��SO42-��
�ʴ�Ϊ��H+��NO3-��Ba2+��Cu2+��I-��S2-��SO42-��K+��
��2����Һ������������������ų�����Ϊ��������ϡ����֮��ķ�Ӧ����Fe+4H++NO3-=Fe3++NO��+2H2O���ʴ�Ϊ��Fe+4H++NO3-=Fe3++NO��+2H2O��
��ȡ��Һ������������������ų������������ӣ����ܺ���NO3-��
����ȡԭ��Һ��Na2SO3��Һ��������ų����������Ƕ�������Ҳ�а�ɫ�������ɣ��ټ������������ȫ��ʧ�����Գ��������ᱵ��һ�����б����ӣ�
һ����������������ӣ�һ��������������ӣ�����ȡԭ��Һ����AgNO3��Һ�������ɣ���ԭ����Һһ������I-��S2-��SO42-��
���Ͽ�֪����Һ��һ������H+��NO3-��Ba2+�����ܺ��е�������K+��һ������Cu2+��I-��S2-��SO42-��
�ʴ�Ϊ��H+��NO3-��Ba2+��Cu2+��I-��S2-��SO42-��K+��
��2����Һ������������������ų�����Ϊ��������ϡ����֮��ķ�Ӧ����Fe+4H++NO3-=Fe3++NO��+2H2O���ʴ�Ϊ��Fe+4H++NO3-=Fe3++NO��+2H2O��
���������⿼�����ӵļ�����ƶϣ���Ŀ�Ѷ��еȣ�ע��������ӷ�Ӧ��ʵ�������ǹؼ���

��ϰ��ϵ�д�
 �ƸԿ�����ҵ��ϵ�д�
�ƸԿ�����ҵ��ϵ�д� ��Ԫ����ĩ��ϰ�ȷ��ϵ�д�
��Ԫ����ĩ��ϰ�ȷ��ϵ�д�
�����Ŀ
���й���Ԫ�ص縺�Դ�С�ıȽ��У�����ȷ���ǣ�������
| A��Se��Te��S��O |
| B��C��N��O��F |
| C��P��S��O��F |
| D��K��Na��Mg��Al |
���з����У�ԭ�ӵ��������Ӳ��ܶ�����8�����ȶ��ṹ���ǣ�������
| A��CO2 |
| B��Cl2 |
| C��H2O |
| D��PF3 |
�����й�ʵ���˵����ȷ���ǣ�������
| A����NH3?H2O��Һ�ܼ���Al3+��Mg2+��Ag+��Cu2+ |
| B������һ�����ʵ���Ũ�ȵ�ϡH2SO4ʱ����Ͳ��ȡŨ��Һʱ�����Ӷ��������ƫ�� |
C����ͼʵ�� ���رյ��ɼУ�������ס��ƿ���������¶�������ð����˵������������ ���رյ��ɼУ�������ס��ƿ���������¶�������ð����˵������������ |
| D���ⶨ�к���ʱ��ȡ25 mL 10 mol/L��Ũ�����25 mL 10 mol/L��NaOH��Һ�кͣ��ⶨ��Ӧ�ų������� |
����ʵ���У���������������ȫ�ܽ���ǣ�������
| A����H2O2��Һ�м�������MnO2��ĩ |
| B����п��Ƥ����������NaOH��Һ�� |
| C��1molͭƬ�뺬2mol H2SO4��Ũ���Ṳ�� |
| D�������£�1molͭƬͶ�뺬4mol HNO3��Ũ������ |
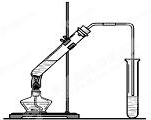 �����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ��������ͼ��ʾ��װ����ȡ�����������ش��������⣺
�����dzµ��㡱��������Ϊ���ڴ������������������ζ��������������ʵ��������Ҳ��������ͼ��ʾ��װ����ȡ�����������ش��������⣺