��Ŀ����
20����0.8mol I2��g����1.2mol H2��g������ij1L�ܱ������У���һ���¶��·�����Ӧ��I2��g��+H2��g��=2HI��g�����ﵽƽ�⣮HI�����������ʱ��ı仯�������ʾ��| HI������� | 1min | 2min | 3min | 4min | 5min | 6min | 7min |
| ����I | 26% | 42% | 52% | 57% | 60% | 60% | 60% |
| ����II | 20% | 33% | 43% | 52% | 57% | 65% | 65% |
��2��������I�ӿ�ʼ��Ӧ������ƽ��ʱ��H2�ķ�Ӧ����Ϊ0.12 mol/��L•min����
��3��Ϊ�ﵽ����II�����ݣ����ڷ�Ӧ��ϵ���ܸı�IJ����ǽ����¶ȣ�
��4���÷�Ӧ�ġ�H��0�����������������=����
��5��������I�´ﵽƽ�����7minʱ���������ѹ��Ϊԭ����һ�룮����ͼ�л���c��HI����ʱ��仯�����ߣ�
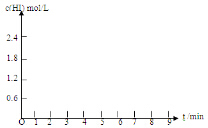
���� ��1���ɱ������ݿ�֪������I��5minʱ����ƽ��״̬����I2����Ũ��Ϊxmol/L����
I2��g��+H2��g��?2HI��g��
��ʼŨ�ȣ�mol/L����0.8 1.2 0
ת��Ũ�ȣ�mol/L����x x 2x
ƽ��Ũ�ȣ�mol/L����0.8-x 1.2-x 2x
����HI����������з��̼���x���ٸ���K=$\frac{{c}^{2}��HI��}{c��{I}_{2}����c��{H}_{2}��}$���㣻
��2������v=$\frac{��c}{��t}$���㣻
��3����ͬʱ����HI�����������С��˵����Ӧ���ʼ�����ƽ��ʱHI���������������Iʱ���ʸı�����ƽ�������ƶ�������ѹǿ��������Ӱ��ƽ���ƶ��������ǽ����¶ȣ�
��4�������¶�ƽ��ʱ�����ƶ���˵������ӦΪ���ȷ�Ӧ��
��5��������I�´ﵽƽ�����7minʱ���������ѹ��Ϊԭ����һ�룬ѹǿ����ƽ�ⲻ�ƶ���HI��Ũ�ȱ�Ϊԭƽ���2����
��� �⣺��1���ɱ������ݿ�֪������I��5minʱ����ƽ��״̬����I2����Ũ��Ϊxmol/L����
I2��g��+H2��g��?2HI��g��
��ʼŨ�ȣ�mol/L����0.8 1.2 0
ת��Ũ�ȣ�mol/L����x x 2x
ƽ��Ũ�ȣ�mol/L����0.8-x 1.2-x 2x
HI���������Ϊ60%����$\frac{2x}{2}$=60%����x=0.6��ƽ�ⳣ��K=$\frac{{c}^{2}��HI��}{c��{I}_{2}����c��{H}_{2}��}$=$\frac{1��{2}^{2}}{0.2��0.6}$=12��
�ʴ�Ϊ��12��
��2��������I�ӿ�ʼ��Ӧ������ƽ��ʱ��H2�ķ�Ӧ����Ϊ$\frac{0.6mol/L}{5min}$=0.12 mol/��L•min����
�ʴ�Ϊ��0.12 mol/��L•min����
��3����ͬʱ����HI�����������С��˵����Ӧ���ʼ�����ƽ��ʱHI���������������Iʱ���ʸı�����ƽ�������ƶ�������ѹǿ��������Ӱ��ƽ���ƶ��������ǽ����¶ȣ�
�ʴ�Ϊ�������¶ȣ�
��4�������¶�ƽ��ʱ�����ƶ���˵������ӦΪ���ȷ�Ӧ������H��0��
�ʴ�Ϊ������
��5��������I�´ﵽƽ���HI��Ũ��Ϊ1.2mol/L����7minʱ���������ѹ��Ϊԭ����һ�룬ѹǿ����ƽ�ⲻ�ƶ���HI��Ũ�ȱ�Ϊԭƽ���2������HIŨ�ȱ�Ϊ2.4mol/L��c��HI����ʱ��仯������Ϊ��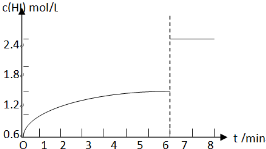 ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼�黯ѧƽ�������Ӱ�����ء���Ӧ���ʼ��㡢ƽ�ⳣ���ȣ�ע��ӣ�3���з�Ӧ������ƽ���ƶ��жϸı�����������ȷ����4�����ʱ䣮

 ���б�ˢ��ϵ�д�
���б�ˢ��ϵ�д� ����кͷ�Ӧ����ѧ��ѧһ����Ҫ�ķ�Ӧ��ijѧ��ʵ��С��Կα��е���������к�ʵ��������о���
����кͷ�Ӧ����ѧ��ѧһ����Ҫ�ķ�Ӧ��ijѧ��ʵ��С��Կα��е���������к�ʵ��������о�����1����ͼ��װ�ý���ǿ�ᣨ50mL 0.5mol/L��HCl����ǿ�50mL 0.55mol/L��NaOH����Ӧ���к��Ȳⶨ��
�ٴ�ʵ��װ���Ͽ���ͼ����ȱ�ٵ�һ�ֲ��������ǻ��β�����������
�ڴ�С�ձ�֮������ֽм�������DZ��¡����ȡ�����ʵ������е�����ɢʧ��
�����ô��������������ʵ�飬ʵ���õ��к��ȵ���ֵƫ�ͣ��ƫ�ߡ�����ƫ�͡�����Ӱ�족����
��2����ʵ����ȷ���Ƶ�0.100mol/L��NaOH��Һ�ⶨijδ֪Ũ�ȵ�ϡ���ᣮ�����ʵ�鲽�����£�
��Ӧѡ��b�ζ��ܣ���дͼ���ж�Ӧ��ĸ������������NaOH��Һ��ϴ2��3��ˮϴ��ĸõζ��ܣ��ټ������0.100mol/L��NaOH��Һ����¼Һ��̶ȶ�����
������ʽ�ζ��ܾ�ȷ�ķų�25.00mL�������ᣬ����������ˮϴ������ƿ�У��ټ���2�η�̪��Һ��
�۵ζ�ʱ���ߵα���ͬʱ�۾�ע����ƿ����Һ��ɫ�ı仯���۲쵽��ƿ����Һ����ɫ����ɫ��Ϊdz��ɫ���Ұ���Ӳ���ɫ���ﵽ�ζ��յ㣻
�ܼ�¼Һ��̶ȶ��������ݵζ��ܵ����ζ����ó����ı�NaOH��Һ����������ظ��ⶨ���Σ�ʵ������¼���±���
| ʵ����� | ��ʼ�ζ��ܶ��� | �յ�ζ��ܶ��� |
| 1 | 0.00mL | 24.02mL |
| 2 | 0.50mL | 24.46mL |
| 3 | 2.50mL | 25.02mL |
��1������ʵ���з�����Ӧ�Ļ�ѧ����ʽ��CuSO4+Zn=ZnSO4+Cu��Zn+H2SO4=ZnSO4+H2����
��2����������ͭ��Һ����Լӿ������������ʵ�ԭ����CuSO4��Zn��Ӧ������Cu��Zn�γ�Cu-Znԭ��أ��ӿ����������ɵ����ʣ�
��3��ʵ����������Na2SO4��MgSO4��Ag2SO4��K2SO4��4����Һ����������ʵ����CuSO4��Һ���������õ���Ag2SO4��
��4��Ҫ�ӿ�����ʵ���������������ʣ����ɲ�ȡ�Ĵ�ʩ�����£�����H2SO4Ũ�� �������ּ��ɣ���
��5��Ϊ�˽�һ���о�����ͭ�����������������ʵ�Ӱ�죬��ͬѧ��������һϵ�е�ʵ�飺�����������Ļ����Һ�ֱ���뵽6��ʢ�й���Zn���������У��ռ����������壬��¼�����ͬ�������������ʱ�䣮
ʵ�� �����Һ | A | B | C | D | E | F |
| 4mol/L H2SO4��mL�� | 30 | V1 | V2 | V3 | V4 | V5 |
| ����CuSO4��Һ��mL�� | 0 | 0.5 | 2.5 | 5 | V6 | 20 |
| H2O��mL�� | V7 | V8 | V9 | V10 | 10 | 0 |
�ڸ�ͬѧ���ó��Ľ���Ϊ������������CuSO4��Һʱ���������������ʻ�����ߣ����������CuSO4��Һ����һ����ʱ���������������ʷ������½���������������������½�����Ҫԭ����һ������CuSO4�����ɵĵ���Cu�������Zn�ı��棬������Zn��H2SO4��Һ�ĽӴ������
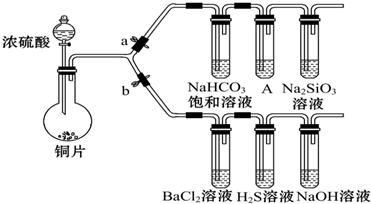
��1����֤SO2�������ԡ���ԭ�Ժ������������ͨ�ԣ�
����������SO2ͨ��NaOH��Һ�У������ӷ���ʽ�ǣ�SO2+NaOH�TNaHSO3
�ڴ�b���ر�a������֤SO2���������ԵĻ�ѧ����ʽ�ǣ�2H2S+SO2�T3S��+2H2O��
��BaCl2��Һ������������ֳ����ݣ��ֱ�μ�������Һ���������ij����Ļ�ѧʽ�����±���Ӧλ�ã�
| �μӵ���Һ | ��ˮ | ��ˮ |
| �����Ļ�ѧʽ |
��2����֤̼����ǽ����Ե����ǿ��������֪���ԣ������̼�ᣩ
��ͭ��Ũ���ᷴӦ�Ļ�ѧ����ʽ��Cu+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CuSO4+SO2��+2H2O��װ��A�е��Լ�������KMnO4��Һ��
����������������װ�õ������ԡ���ҩƷ��a�ر�b��Ȼ�����Ũ���ᣬ���ȣ�
����˵��̼�ķǽ����Աȹ�ǿ��ʵ�������ǣ�A������KMnO4��Һ����ɫ��Na2SiO3��Һ�г��ְ�ɫ������
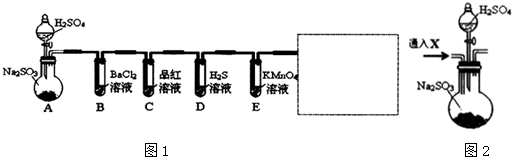
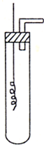 ijѧϰС��Ϊ֤�����۲�ͭ��ϡHNO3��Ӧ�IJ�����NO�����������ͼ��ʾ��ʵ��װ�ã�����������ǵ�˼·��ѡ������ҩƷ���ʵ�飬��дʵ�鲽�貢�ش����⣮��ѡ���ҩƷ��ͭ˿��ϡ���ᡢ�ռ���Һ�����ۡ�CaCO3������
ijѧϰС��Ϊ֤�����۲�ͭ��ϡHNO3��Ӧ�IJ�����NO�����������ͼ��ʾ��ʵ��װ�ã�����������ǵ�˼·��ѡ������ҩƷ���ʵ�飬��дʵ�鲽�貢�ش����⣮��ѡ���ҩƷ��ͭ˿��ϡ���ᡢ�ռ���Һ�����ۡ�CaCO3������ ��
�� ��
�� ��
��