��Ŀ����
12��A��B��C��DΪ������ˮ�����ֹ��壬B��ˮ��Һ��dz��ɫ��DΪ�������Σ�������ǵ������У�| ������ | Fe2+��Na+��Ba2+��Al3+ |
| ������ | Cl-��SO42-��NO3-��OH- |
��1��A��Һ����ɫ��ӦΪ��ɫ����A��Һ�к��еĽ���������Na+����������ţ���
��2����A��ˮ��Һ��μ���C�У������ɰ�ɫ���������ɫ�������ܽ⣬��д����ɫ�������ܽ�����ӷ���ʽAl��OH��3+OH-=AlO2-+2H2O��
��3��B��Һ��D��Һ��Ӧ���ɲ�����ϡ����İ�ɫ��������д���÷�Ӧ�����ӷ���ʽBa2++SO42-=BaSO4����
��4���ݴ��ƶ������ǣ�д��ѧʽ��ANaOH��DBa��NO3
��2��
��5��A��Һ��B��Һ��Ӧ������Ϊ�����ɰ�ɫ��״������Ȼ���Ϊ����ɫ������Ϊ���ɫ��
���� A��B��C��DΪ������ˮ�����ֹ��壬B��ˮ��Һ��dz��ɫ��˵��B�к���Fe2+��DΪ�������Σ������ݣ�3����֪��BΪ�����Σ���D�к����������
��1�����Լ����������ɫ��ӦΪ��ɫ��
��2������������ֻ�����������������������ɵ����������ܼ����ܽ��ڼ��Է�Ӧ�У��ݴ˽��
��3����ɫ������ϡ����ij���Ϊ���ᱵ���ݴ���д���ӷ���ʽ��
��4�����ݷ����شɣ�
��5����������������������Ӧ������ش�
��� �⣺���ݷ�����֪��A��һ�����������ӣ��ҽ�A����Һ����C�У������ɰ�ɫ����������ܽ⣬˵��A��NaOH��ֻ��B��Һ����ɫ��˵��B�к���Fe2+����B��F��Ӧ��������������İ�ɫ������˵��B����������D�����ᱵ��
��1���Ƶ����Լ����������ɫ��ӦΪ��ɫ����A��Һ�к��еĽ��������������ӣ��ʴ�Ϊ��Na+��
��2����A��ˮ��Һ��μ���C�У������ɰ�ɫ���������ɫ�������ܽ⣬˵���˳����������������������������������ӷ�Ӧ����ƫ���������ɫ�������ܽ�����ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O���ʴ�Ϊ��Al��OH��3+OH-=AlO2-+2H2O��
��3�������������η�Ӧ�������ᱵ�������ó���������ϡ���ᣬ���ӷ�Ӧ����ʽΪ��Ba2++SO42-=BaSO4�����ʴ�Ϊ��Ba2++SO42-=BaSO4����
��4�����ݷ�����֪A��NaOH��D�����ᱵ���ʴ�Ϊ��NaOH��Ba��NO3��2��
��5����������������������Ӧ�������ɰ�ɫ��״������Ȼ���Ϊ����ɫ������Ϊ���ɫ���ʴ�Ϊ�������ɰ�ɫ��״������Ȼ���Ϊ����ɫ������Ϊ���ɫ��
���� ���⿼�������ӹ�������жϣ�ע���������ʵ�Ӧ�ã���Ӧ������ж��ǽ���ؼ��������Ѷ��еȣ�

 ��ѧ��������������Ͼ���ѧ������ϵ�д�
��ѧ��������������Ͼ���ѧ������ϵ�д�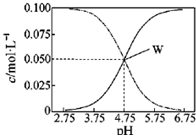 25�棬��c��CH3COOH��+c��CH3COO-��=0��l mol/L��һ�����ʹ����ƻ����Һ����Һ��c��CH3COOH����c��CH3COO-����pHֵ�Ĺ�ϵ��ͼ��ʾ���й�����Ũ�ȹ�ϵ������ȷ���ǣ�������
25�棬��c��CH3COOH��+c��CH3COO-��=0��l mol/L��һ�����ʹ����ƻ����Һ����Һ��c��CH3COOH����c��CH3COO-����pHֵ�Ĺ�ϵ��ͼ��ʾ���й�����Ũ�ȹ�ϵ������ȷ���ǣ�������| A�� | pH=5.5��Һ�У�c��CH3COO-��c��CH3COOH����c��H+����c��OHһ�� | |
| B�� | W���ʾ��Һ�У�c��Na+��=c��CH3COO-�� | |
| C�� | pH=3.5��Һ�У�c��Na+��-c��OH-��+c��CH3COOH��=0��l moL/L | |
| D�� | ��W������ʾ��Һ��ͨ��0.05molHCl���壨��Һ����仯�ɺ��ԣ���c��H+��=c��CH3COOH��+c��OH-�� |
| A�� | ����p% | B�� | ����p% | C�� | С��p | D�� | ���ж� |
| A�� | ϡ��ǰ����ҺpH���٣���=�� | |
| B�� | ϡ�ͺ���ҺpH���٣���=�� | |
| C�� | ϡ��ǰ����Һ�����ʵ����ʵ���Ũ�ȣ��ۣ���=�� | |
| D�� | ϡ�ͺ���Һ�����ʵ����ʵ���Ũ�ȣ��ۣ���=�� |
| ������ | CaF2 | CaC2O4 |
| Ksp | 3.5��10-11 | 2.3��10-9 |
| �� | HF | H2C2O4 |
| ���볣�� | Ka=7.2��10-4 | Ka1=5.4��10-2 Ka2=5.4��10-5 |
��2����0.1mol•L-1�IJ��������Һ�еμ�NaOH��Һ����Һ�����ԣ���ʱ��Һ�и����ӵ�Ũ�ȹ�ϵ��ȷ����ad������ĸ����
a��c��K+��=c��HC2O4-��+c��H2C2O4��+c��C2O42-��
b��c��K+��+c��Na+��=c��HC2O4-��+c��C2O42-��
c��cNa+��=c��H2C2O4��+c��C2O42-��
d��c��K+����cNa+��
��3����Ũ��Ϊ1mol•L-1��HF��0.01mol•L-1��KHC2O4-�����Һ����μ���1mol•L-1��CaCl2��Һ������õ��ij���ΪCaF2���ѧʽ����
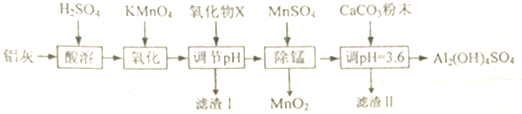
��֪�������������������pH
| Al��OH��3 | Fe��OH��2 | Fe��OH��3 | |
| ��ʼ����ʱ | 3.4 | 6.3 | 1.5 |
| ��ȫ����ʱ | 4.7 | 8.3 | 2.8 |
��2�����һ����ʵ�飬֤��������������Ԫ���ѱ�������ȫȡ��һ�ε���PH�����Һ���Թ��У��μ����軯����Һ������Һ��Ѫ��ɫ��˵����������ȫ����֮������ȫ
��3��д����һ�ε���pH�ķ�Χ[2.8��3.4�����ڶ��ε���pHʱ������Һ��pHƫ�ߣ����ᵼ����Һ����Ԫ�صĺ�������
��4������CaC03�Ʊ���ʽ�������Ļ�ѧ����ʽΪAl2��SO4��3+2CaC03+2H2O=Al2��OH��4SO4+2CaSO4+2C02����
 ʵ������Ҫ0.1mol/L NaOH��Һ450mL����Ҫ0.5mol/L������Һ500mL��������������Һ����������ش��������⣺
ʵ������Ҫ0.1mol/L NaOH��Һ450mL����Ҫ0.5mol/L������Һ500mL��������������Һ����������ش��������⣺