��Ŀ����
(1) ��4�֣��ڴ��л������ܱ������з�����Ӧ��Fe2O3��3H2===2Fe��3H2O����ȡ���д�ʩ���ܸı䷴Ӧ���ʵ���(����)
| A������Fe2O3��Ͷ���� |
| B������������������䣬����H2�������� |
| C������N2������������ѹǿ���� |
| D������N2����������������� |
(2)��H2��Ũ�ȱ仯��ʾ2 s�ڵ�ƽ������v(H2)��________��
(3)������ת������________��
(4)ƽ��ʱ��������а������������________��
(5)�����ڷ�Ӧǰ��ѹǿ����________��
(1)AD(12�֣�ÿ��2�֣���2��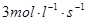 (3) 50% (4)33.3% (5)4:3
(3) 50% (4)33.3% (5)4:3
����

��ϰ��ϵ�д�
 �Ǽ�����������ϵ�д�
�Ǽ�����������ϵ�д�
�����Ŀ