��Ŀ����
��1����һ�������£�RO3n-��I-������Ӧ�����ӷ���ʽ���£�RO3n-+6I-+6H+=R-+3I2+3H2O��
RO3n-��RԪ�صĻ��ϼ��� ����Ӧ����2moL I2����ʱ��ת�Ƶ��ӵ����ʵ���Ϊ ��
��2������淋ĺ�����Ϊ ��ǿ���������ֽܷ⣬���ɰ���������������ˮ��д������ƽ��Ӧ����ʽ�� ��
��Ӧ�����ɵ���������ͻ�ԭ��������ʵ���֮���� ��
RO3n-��RԪ�صĻ��ϼ���
��2������淋ĺ�����Ϊ
��Ӧ�����ɵ���������ͻ�ԭ��������ʵ���֮����
���㣺������ԭ��Ӧ�ļ���,������ԭ��Ӧ
ר�⣺������ԭ��Ӧר��
��������1���������ӷ���ʽ��ѭ����غ��ж�RO3n-����������ɣ����Դ��жϻ��ϼۣ���ϵ��ӵ�ת����Ŀ���㣻
��2����Ӧ�ķ���ʽΪ3��NH4��2SO4=4NH3+3SO2+N2+6H2O��������������ΪN2����ԭ����ΪSO2����Ϸ���ʽ���㣮
��2����Ӧ�ķ���ʽΪ3��NH4��2SO4=4NH3+3SO2+N2+6H2O��������������ΪN2����ԭ����ΪSO2����Ϸ���ʽ���㣮
���
�⣺��1���������ӷ���ʽ��ѭ����غ��֪����-n��+��-6��+6=-1����֪n=1������RO3n-���ӻ��ϼ۴�����ϵ����x-2��3��=-1����R�Ļ��ϼ�Ϊx������֪x=+5����Ӧ��IԪ�ػ��ϼ���-1�����ߵ�0�ۣ���2moL I2����ʱ����ת��4mol���ӣ�
�ʴ�Ϊ��+5��4��
��2����NH4��2SO4�ĺ�����Ϊ
��100%=21.2%����ƽ��ʽΪ��3��NH4��2SO4=4NH3+3SO2+N2+6H2O��������������ΪN2����ԭ����ΪSO2�������ʵ���֮��Ϊ1��3��
�ʴ�Ϊ��21.2%��3��NH4��2SO4=4NH3+3SO2+N2+6H2O��1��3��
�ʴ�Ϊ��+5��4��
��2����NH4��2SO4�ĺ�����Ϊ
| 14��2 |
| 132 |
�ʴ�Ϊ��21.2%��3��NH4��2SO4=4NH3+3SO2+N2+6H2O��1��3��
�����������ۺϿ���������ԭ��Ӧ��Ϊ��Ƶ���㣬������ѧ���ķ��������ͼ��������Ŀ��飬ע����غ�ĽǶȽ����⣬�ѶȲ���

��ϰ��ϵ�д�
 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
�����Ŀ
����ϵͳ��������ȷ���ǣ�������
| A��2-��-4-�һ����� |
| B��2��3-���һ�-1-��ϩ |
| C��2-��-3-��Ȳ |
| D��2���� |
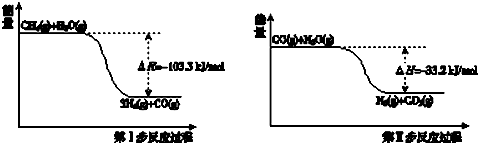
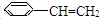 ��5���л���ֱ���һ����������H2��ַ�Ӧ�������и���Ҫ�����
��5���л���ֱ���һ����������H2��ַ�Ӧ�������и���Ҫ�����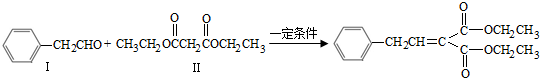
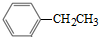 �ɺϳɻ�����I�����������
�ɺϳɻ�����I�����������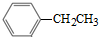 ��ͬ���칹�壬���ĺ˴Ź�������������壬�����֮��Ϊ4��6������CrO-�������·�Ӧ����������������Ӧ�Ļ�����V�����Ľṹ��ʽΪ
��ͬ���칹�壬���ĺ˴Ź�������������壬�����֮��Ϊ4��6������CrO-�������·�Ӧ����������������Ӧ�Ļ�����V�����Ľṹ��ʽΪ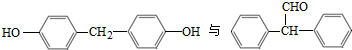 Ҳ���Է������Ʒ�Ӧ�ٵķ�Ӧ������ĽṹʽΪ
Ҳ���Է������Ʒ�Ӧ�ٵķ�Ӧ������ĽṹʽΪ