��Ŀ����
��16�֣�ij��ѧ��ȤС��������з����ⶨNa2SO3��Ʒ��Na2SO4������������
������һ�� �� SO2������������ͼ��ʾװ�ã�ͨ��ʵ��ⶨ����SO2��������
�� SO2������������ͼ��ʾװ�ã�ͨ��ʵ��ⶨ����SO2��������
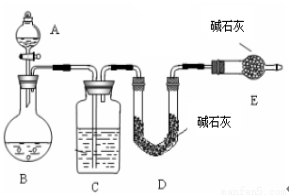
��1��C��ʢ�ŵ��Լ��� ��Eװ�õ������� ��
��2����ʵ��ǰ��ȡm1g��Ʒ���ٲ��SO2������Ϊm2g����ɵ�Na2SO4��������������m2��ͨ��
�ⶨ (��װ�÷���)װ����ʵ��ǰ���������õ��ġ��÷�����ȱ���ǣ�ֻ��һ�㣩
��
���������������������
��һ������ȡm3g��Ʒ������С�ձ��� �ڶ�������С�ձ��м�������ϡ���ᣬ����
������������С�ձ��м�������BaCl2��Һ��Ȼ����ˡ�ϴ�� ���IJ����������������Ϊm4g
��3��������BaCl2�ܷ�ij�Ba(NO3)2�� ����ܡ����ܡ����� �Լ���ԭ��
��
��4������ϴ�ӳ����ķ����� ��
��5����m3=2m4 ��������Na2SO4����������Ϊ ��
��16�֣�ÿ��2�֣�
����һ:
��1��Ũ�����ֹD�м�ʯ�����տ����еĶ�����̼��ˮ����
��2��D SO2��һ����ȫ�����գ�װ�����в���SO2����
������:
��3�����ܣ�����������Ba(NO3)2��SO2������BaSO4
��4����©���ڼ�����ˮ���պý�û��������ˮ��Ȼ�������ټ�ˮϴ��2~3�Ρ�
��5��30.5%
��������
�������������һ����1���������ɵĶ���������������������Ƶ���������������װ��c��ʢ��Ũ��������������װ��E�������Ƿ�ֹD�м�ʯ�����տ����еĶ�����̼��ˮ�������Ӷ�ʹ�����������������ʵ�������
��2��Dװ�õ�����������ֵ��Ϊ�������������������m2��ͨ��װ��D�õ��ģ����÷����IJ���֮���������Ķ�����������ڵ������У�δ����ȫ���գ�
���������÷����Dzⶨ���ᱵ�����������������Ƶ�������������3�����ܸ�Ϊ���ᱵ����Ϊ��Ϊ���ᱵ��Һ���������������¶�������ᱻ�������������ɵij���ȫ�������ᱵ,ʹ���ƫ�ߣ�
��4��ϴ�ӳ����ķ�������©���ڼ�����ˮ���պý�û��������ˮ��Ȼ�������ټ�ˮϴ��2~3�Σ�
��5��m4Ϊ���ᱵ���������������Ƶ�����Ϊm4/233��142g����m3=2m4������Ʒ�������Ƶ���������Ϊm4/233��142/m3��100%=71/233=30.5%��
���㣺�����ʵ��ķ������ۣ����������ļ��㣬������ϴ�Ӳ���

������������һ���������ܹ��棬��������Ӧ�Լ���ᷢ����ѧ�仯�����������ӷ���ʽ��ȷ����
ѡ�� | ������ | �����Լ� | �����Լ�������Ӧ�����ӷ���ʽ |
A | Fe2����NO | ϡ���� | 3Fe 2����NO |
B | Fe3����I����ClO�� | ����������Һ | Fe3����3OH��===Fe(OH)3�� |
C | Ba2����HCO | ����������Һ | HCO |
D | Al3����Cl����NO | ��������������Һ | Al3����3OH��===Al(OH)3�� |
 5Ca2+(aq)��3PO43-(aq)��OH- (aq) Ksp = 6��8��10-37
5Ca2+(aq)��3PO43-(aq)��OH- (aq) Ksp = 6��8��10-37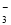 ��K��
��K��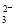 ��H2O
��H2O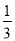 �����ֳ��Թܼг���ĩ�ˣ����м���
�����ֳ��Թܼг���ĩ�ˣ����м���