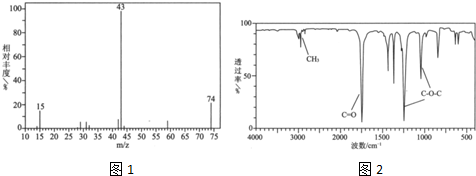
��Ŀ����
������C��H��O���л���3.24gװ��Ԫ�ط���װ�ã�ͨ��������O2ʹ֮��ȫȼ�գ������ɵ���������ͨ��װ��Ũ�����ϴ��ƿ�ͼ�ʯ�ҵĸ���ܣ����ϴ��ƿ������������2.16g������ܵ�����������9.24g����֪���л������Է�������Ϊ108��
��1������л���ķ���ʽ��
��2����֪�������к��б�������1mol������������Ʒ�Ӧ��������״��������0.5mol����д�������ʵ���һ�ֽṹ��ʽ��
��3��ȼ�մ��л���3.24g������O2 �ˣ�
��1������л���ķ���ʽ��
��2����֪�������к��б�������1mol������������Ʒ�Ӧ��������״��������0.5mol����д�������ʵ���һ�ֽṹ��ʽ��
��3��ȼ�մ��л���3.24g������O2
���㣺�й��л������ʽȷ���ļ���
ר�⣺�������������ȼ�չ���
��������1������ϴ��ƿ������ܵ������仯�ֱ���������ˮ��������̼�����������ʵ������ٸ��������غ�����3.24g���л�������к��е���Ԫ�ص����������ʵ�����������������ʽ��
��2�������л���ķ���ʽ�������Ϣ�ж��京�еĹ����ţ�Ȼ��д������ܵĽṹ��ʽ��
��3��3.24g���л���ȼ����������������=���ɶ�����̼������+����ˮ������-�л����������
��2�������л���ķ���ʽ�������Ϣ�ж��京�еĹ����ţ�Ȼ��д������ܵĽṹ��ʽ��
��3��3.24g���л���ȼ����������������=���ɶ�����̼������+����ˮ������-�л����������
���
�⣺���л�����ȫȼ�ղ���ΪH2O��CO2��װ��Ũ�����ϴ��ƿ���ӵ���ˮ��������ˮ�����ʵ���Ϊ��n��H2O��=
=0.12mol����n��H��=0.12��2=0.24mol��
װ�м�ʯ�ҵĸ�������ӵ���CO2��������������̼�����ʵ���Ϊ��n��CO2��=
=0.21mol����n��C��=0.21mol��
���л�������Ԫ�ص�����Ϊ��m��O��=3.24-0.24-2.52=0.48g����Ԫ�ص����ʵ���Ϊ��n��O��=
=0.03mol��
��1��3.24g���л�������ʵ���Ϊ��n���л��=
=0.03mol��
��n������n��C����n��H����n��O��=0.03��0.21��0.24��0.03=1��7��8��1��
�ʸ��л���ķ���ʽΪ��C7H8O��
�𣺸��л���ķ���ʽΪC7H8O��
��2����֪�������к��б�������1mol������������Ʒ�Ӧ��������״��������0.5mol��˵�������к���1��-OH����ṹ�����DZ��״�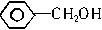 ���ڼ�����
���ڼ����� ���������
���������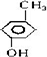 ���Լ�����
���Լ����� ��
��
�ʴ�Ϊ��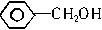 ����
����  ��
�� ��
�� ��
��
��3�����ĵ��������������ڷ�Ӧ����������-�л��������������m��O2��=m��H2O��+m��CO2��-m��C7H8O��=2.16g+9.24g-3.24g=8.16g��
�ʴ�Ϊ��8.16��
| 2.16g |
| 18g/mol |
װ�м�ʯ�ҵĸ�������ӵ���CO2��������������̼�����ʵ���Ϊ��n��CO2��=
| 9.24g |
| 44g/mol |
���л�������Ԫ�ص�����Ϊ��m��O��=3.24-0.24-2.52=0.48g����Ԫ�ص����ʵ���Ϊ��n��O��=
| 0.48g |
| 16g/mol |
��1��3.24g���л�������ʵ���Ϊ��n���л��=
| 3.24g |
| 108g/mol |
��n������n��C����n��H����n��O��=0.03��0.21��0.24��0.03=1��7��8��1��
�ʸ��л���ķ���ʽΪ��C7H8O��
�𣺸��л���ķ���ʽΪC7H8O��
��2����֪�������к��б�������1mol������������Ʒ�Ӧ��������״��������0.5mol��˵�������к���1��-OH����ṹ�����DZ��״�
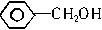 ���ڼ�����
���ڼ����� ���������
��������� ���Լ�����
���Լ����� ��
���ʴ�Ϊ��
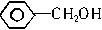 ����
����  ��
�� ��
�� ��
����3�����ĵ��������������ڷ�Ӧ����������-�л��������������m��O2��=m��H2O��+m��CO2��-m��C7H8O��=2.16g+9.24g-3.24g=8.16g��
�ʴ�Ϊ��8.16��
�����������ۺϿ����л������ʽ��ȷ���Լ��л���ṹ���Ƶ�����Ŀ�Ѷ��еȣ�ע������ȷ���л������ʽ���ṹ��ʽ�ķ�������ȷ��3���м����������������������㷽����

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
�������pH��������ȷ���ǣ�������
| A����7 |
| B����5.6 |
| C����5.6��7֮�� |
| D������5.6 |