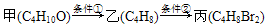��Ŀ����
( 10��) �ס������ص缫���϶���������̼��������ͼ������ش��������⣺

(1)�������о�ʢ��CuSO4��Һ����Ӧһ��ʱ���
���к�ɫ�����������ǣ��׳��е� �����ҳ��е� ����
�����ҳ��������ĵ缫��Ӧʽ�� ��
(2)�������о�ʢ�ű���NaCl��Һ��
��д���ҳ��з����ܷ�Ӧ�����ӷ���ʽ ��
�ڽ�ʪ��ĵ���KI��ֽ�����ҳظ�����������ֽ��������һ��ʱ����ַ�����ɫ��ȥ��������Ϊ������Cl2�����ɵ�I2����������Ӧ��Cl2��I2�����ʵ���֮��Ϊ5:1�������������ᡣ�÷�Ӧ�Ļ�ѧ����ʽΪ ��
�����ҳ�ת��0.02mol���Ӻ�ֹͣʵ�飬������Һ�������200mL������Һ���Ⱥ��pH= ��
(1)�� ̼(C )����(Fe) (2��)
�� 4 OH��-4 e��=== 2H2O + O2�� (2��)
(2)��2 Cl��+2H2O![]() 2 OH��+H2��+Cl2�� (2��)
2 OH��+H2��+Cl2�� (2��)
��5Cl2 + I2 + 6H2O === 10HCl + 2HIO3 (2��)
��13 (2��)
����:
���⿼��绯����ԭ�����йؼ��㣻��1����ͼ��֪����Ϊԭ��أ���Ϊ������̼��Ϊ������ͭ������̼���ϱ���ԭ���ɿ�����ɫ������������Ϊ���أ����ݵ�������֪̼��Ϊ��������Ϊ��������Ӧһ��ʱ���ͭ��������������ԭ���ɿ����к�ɫͭ�������������ͭ�ǵ��ˮ�����ʣ������缫��ӦʽΪ��4 OH��-4 e��=== 2H2O + O2�� ��2����ⱥ��NaCl��Һ�ܷ�Ӧ�����ӷ���ʽΪ��2 Cl��+2H2O![]() 2 OH��+H2��+Cl2�� ������Ӧ��Cl2��I2�����ʵ���֮��Ϊ5:1�����ݵ�ʧ�����غ㣬��Ӧ�Ļ�ѧ����ʽΪ��5Cl2 + I2 + 6H2O === 10HCl + 2HIO3 ��
2 OH��+H2��+Cl2�� ������Ӧ��Cl2��I2�����ʵ���֮��Ϊ5:1�����ݵ�ʧ�����غ㣬��Ӧ�Ļ�ѧ����ʽΪ��5Cl2 + I2 + 6H2O === 10HCl + 2HIO3 ��
2 Cl��+2H2O![]() 2 OH��+H2��+Cl2�� ת�Ƶ���
2 OH��+H2��+Cl2�� ת�Ƶ���
2 2 mol
n(OH��) 0.02mol
n(OH��) =0.02mol
c (OH��)= =0.1mol/L
pH=13

 xC(g)�ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2���Իش������й����⣺
xC(g)�ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2���Իش������й����⣺ xC(g)�ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2���Իش������й����⣺
xC(g)�ﵽƽ�⣬��ʱ��üס�����������C�����������Ϊ0.2���Իش������й����⣺