��Ŀ����
��12�֣������ҹ�������Դ���������ѵ����ֲ�ʾ��ͼ����ͼ����ĸ������Դ����������������ס��ҡ���Ϊ��Դ�����������ͷ��ʾ��Դ���ͷ���

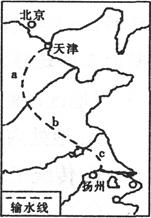
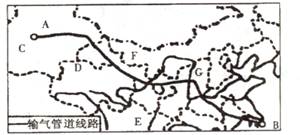
(1)ͼ����Ͽˮ������λ�� ����д��ĸ�����õ���ˮ����Դ��Ϊ�ḻ��ԭ���� ��
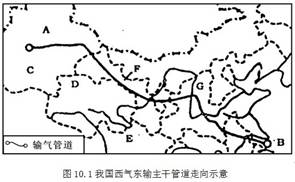
��2��ͼ�������ҹ��ص㿪����ʯ�͡���Ȼ����Դ������ ����д��ĸ�����ɸû������ҵ�������Դ������Ϊ ���̣���B��C��D��R���������ס��ҡ�����������繤�̱���Ϊ ���̡�������Щ��Դ���̵���Ҫ�������� ��ѡ����գ���
A�����ⶫ��������Դ���ѽ���״��
B������������������Դ��Ӧ״��
C���������������Ļ�����Ⱦ
D������������������Դ���ѽṹ
(3) ��ͼ��ӳ�ҹ���Դ��������Դ���ѵķֲ��ص��� ��Ϊ�����ҹ���Դ��Ӧ�������⣬����������������Դ�����⣬����Ϊ�����Բ�ȡ��Щ��Ҫ��ʩ��
��

(1)ͼ����Ͽˮ������λ�� ����д��ĸ�����õ���ˮ����Դ��Ϊ�ḻ��ԭ���� ��
��2��ͼ�������ҹ��ص㿪����ʯ�͡���Ȼ����Դ������ ����д��ĸ�����ɸû������ҵ�������Դ������Ϊ ���̣���B��C��D��R���������ס��ҡ�����������繤�̱���Ϊ ���̡�������Щ��Դ���̵���Ҫ�������� ��ѡ����գ���
A�����ⶫ��������Դ���ѽ���״��
B������������������Դ��Ӧ״��
C���������������Ļ�����Ⱦ
D������������������Դ���ѽṹ
(3) ��ͼ��ӳ�ҹ���Դ��������Դ���ѵķֲ��ص��� ��Ϊ�����ҹ���Դ��Ӧ�������⣬����������������Դ�����⣬����Ϊ�����Բ�ȡ��Щ��Ҫ��ʩ��
��
��1��R ˮ����ˮ�������� ��3�֣�
��2��A �������� ���綫�� A D ��4�֣�
��3����Դ���������ѵ����ֲ��ܲ�ƽ�⣨�����Դ�����������Ҫ�����������������غ���������Ҫ��Դ���ѵ���������1�֣� ������ҵ�ṹ��ʵ�м������죬������Դ����������Լ��Դ�������˷ѣ�Ŭ��������������Դ��������Դ�ȣ�3�֣�
��2��A �������� ���綫�� A D ��4�֣�
��3����Դ���������ѵ����ֲ��ܲ�ƽ�⣨�����Դ�����������Ҫ�����������������غ���������Ҫ��Դ���ѵ���������1�֣� ������ҵ�ṹ��ʵ�м������죬������Դ����������Լ��Դ�������˷ѣ�Ŭ��������������Դ��������Դ�ȣ�3�֣�
���⿼���ҹ�����Դ��������乤�̡���1����Ͽ����λ�ڳ������ε�����ˮ����Դ�ḻ��Ҫ�Ӻ���������ͺ��������������������2��ͼʾAΪ����ľ��أ����ҹ��ص㿪����ʯ�͡���Ȼ�����ء�������ľ������ҵأ��Ϻ������͵���Դ����Ϊ�������乤�̡�ͼʾ������������������繤��Ϊ���綫���̡��ù��̵�������Ҫ���ڴٽ�������������Դ��Դ�Ŀ���������Դ����Ϊ�������ƣ��ٽ����������ľ��÷�չ�����Զ��������������ǣ����ⶫ����������Դ��Ӧ�������ڸ��ƶ�����������Դ���ѽṹ�����ᶫ�������Ĵ�����Ⱦ�����ƴ���������������ѡAD���3���ҹ�����Դ������Ҫ�ֲ����������������ҹ�����Դ�����Զ�������Ϊ��������Դ���͡��������ǻ����ҹ���Դ�������Ҫ;����

��ϰ��ϵ�д�
 ����5��2���ϵ�д�
����5��2���ϵ�д�
�����Ŀ