��Ŀ����
4��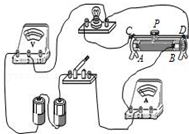 С��ͬѧ����2.5VС���ݵĵ���ʱ�����ӵĵ�·��ͼ��ʾ��
С��ͬѧ����2.5VС���ݵĵ���ʱ�����ӵĵ�·��ͼ��ʾ����1������·��������һ���������Ӵ������������Ӵ���ĵ����ϴ�������û�и������պϿ��أ���ѹ��ָ��ƫת�Ƕȹ����п��ܱ��ջ���
��2������ͼ�в�������ȷ�����ߣ�
��3������������ڱպϿ���ǰ��С��Ӧ�������������Ļ�Ƭ����A�ˣ���ѡ�A����B����
��4��С�ո����������ȷ�IJ�����õ���������ӱ��м����֪����ʵ��С���ݵĵ��費��ȣ�����Ϊԭ������ǵ����ڲ�ͬ��ѹ�µ�˿�¶Ȳ�ͬ����˿�ĵ������¶ȵ����߶�����
| ʵ����� | 1 | 2 | 3 |
| ��ѹU/V | 2.0 | 2.5 | 2.8 |
| ����I/A | 0.20 | 0.24 | 0.25 |
���� ��1������·���ӵĴ�����Ҫ�ӵ���������ѹ�������ӷ��������̡������������������������ӷ��ȷ��濼�ǣ�
��2�������·�Ĵ���Ҫͨ��һ�����߸������������Ҽ�˵�ѹ�����������ݲ���·������������һ��һ�£�
��3�����ӵ�·��������ʶ��ע��������ӵ�·ʱ���ضϿ������������Ӵ����µĶ�·�����ջ�Ԫ�����պϿ���ǰ����������������ֵ�������������·��
��4����˿�ĵ������¶�Ӱ��ϴ��¶ȱ仯ʱ����˿�ĵ���Ҫ�����仯��
��� �⣺��1��������е�·����ѹ�������ڵ�·���ˣ����ݱ���·�ˣ����ӵƵ������������������������֮��ĵ����Ǵ���ģ���ͼ1��ʾ��
��ѹ�������ڵ�·�У��൱�ڲ�����Դ��ѹ�����ѹ��ѡ��С���̣���ѹ��ָ��ƫת�Ƕȹ����п����ջ���
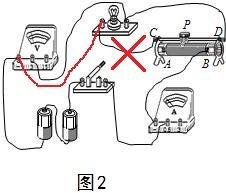
��2�������ӵ�����������������������������ȥ�������Ƶ�����������ѹ�����������֮��������������ͼ2��ʾ��
��3������������ʹ�õ������½��������պϿ���ǰҪʹ�������ֵ��ʽ���Ƭ����A�ˣ�
��4������R=$\frac{U}{I}$�����֪��˿��ֵ���Ƕ�ֵ�����β����õ��ĵ���ֵԽ��Խ��������Ϊ�������˵�ѹ����ʱ��ͨ���ĵ���ҲԽ��Խ����P=UI���ƵĹ���ҲԽ��Խ��˿���¶������ˣ�����˿�ĵ������¶ȵ����߶�����
�ʴ�Ϊ��
��1����ͼ1��ʾ�� ָ��ƫת�Ƕȹ����п��ܱ��ջ���
��2����ͼ2��ʾ��
��3��A��
��4�������ڲ�ͬ��ѹ�µ�˿�¶Ȳ�ͬ����˿�ĵ������¶ȵ����߶�����
���� �����÷���������裬�����·�����ӡ�ʵ��ע�����Ӱ������С�����ص�֪ʶ���Ѷ�һ�㣮

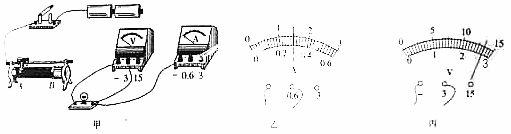
��1�������ñʻ��ߴ��浼�ߣ���ʵ���·����������Ҫ���������Ļ�ƬP��B�ƶ���������ʾ���������߲������森
��2������Ʊ�ʵ������ݱ���
| ʵ����� | |||
| 1 | |||
| 2 | |||
| 3 |
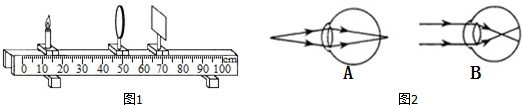
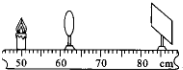 ���ý���Ϊ10cm������̽��������ɵ�ʵ���У�
���ý���Ϊ10cm������̽��������ɵ�ʵ���У�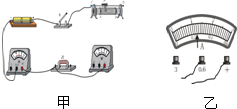 ijͬѧ���õ���������ѹ������һ�ε���˿R�ĵ��裬���������˲��ֵ�·����ͼ����ʾ���������������в��裺
ijͬѧ���õ���������ѹ������һ�ε���˿R�ĵ��裬���������˲��ֵ�·����ͼ����ʾ���������������в��裺 �ü�ͷ����ż���Χ�Ÿ��ߵķ���
�ü�ͷ����ż���Χ�Ÿ��ߵķ���