��Ŀ����
6��������ijС��̽��ͨ����������뵼������ϵ��ʵ�飬ʵ�������õĶ�ֵ����ֱ���5����10����15����20�������������������֣���20�� 2A���͡�50�� 1A������Դ��ѹΪ4.5V����ش��������⣮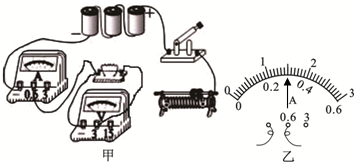
��1������ͼ�����ñʻ��ߴ��浼�߽���·����������Ҫ��ʵ�����������Ƭ���������ƶ���������ʾ�����
��2����ȷ���ӵ�·�պϿ��أ����ֵ�ѹ����ʾ����������û��ʾ�����������ֹ��ϵ�ԭ������Ƕ�ֵ�����·����֪��������ã���
��3���ų����Ϻ���ij�β����У���5���Ķ�ֵ���������·ʱ�����ڻ�����������ʹ��ѹ����ʾ��Ϊ1.5V����ʱ��������ʾ����ͼ�ң���������Դ�ĵ���Ϊ0.3A����ʱ��ֵ����ĵ繦����0.45W��
��4�����Ű�5�����軻��10���ĵ��裬Ӧ�����ң�ѡ������ҡ����ƶ������������Ļ�����ͷ��ʹ��ѹ����ʾ��Ϊ1.5V���ټ��µ���ֵ��
��5��Ϊ�������ṩ��4����ֵ������ʵ���ж������ϣ�Ӧ��ѡ����Ϊ��50�� 1A���Ļ�����������
���� ��1������ʵ���л�Ƭ���������ƶ���������ʾ�����ȷ�������������ӷ�ʽ��
��2�����ֵ�ѹ����ʾ����˵����ѹ�����Դ��ͨ��������û��ʾ����˵����·��·���·���ܵ���ܴݴ��жϣ�
��3�����������С���̵ķֶ�ֵ����������P=UI��繦�ʣ�
��4�����ݷ�ѹԭ����ȷ����Ƭ�ƶ��ķ���
��5������ֵ����Ϊ20��ʱ�����ݷ�ѹԭ����������������·�е���С��ֵȷ���������Ĺ��
��� 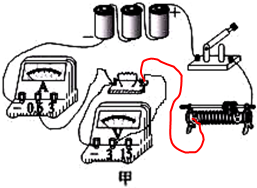 �⣺��1������Ƭ���������ƶ��������������·�еĵ����С���ʱ�������Ƭ���ҵ���˿�����·������ͼ��ʾ��
�⣺��1������Ƭ���������ƶ��������������·�еĵ����С���ʱ�������Ƭ���ҵ���˿�����·������ͼ��ʾ��
��2���պϿ��أ����ֵ�ѹ����ʾ����˵����ѹ�����Դ��ͨ��������û��ʾ����˵����·��·���·���ܵ���ܴ����ԣ��������ֹ��ϵ�ԭ������Ƕ�ֵ�����·�����µ�ѹ�������ڵ�·�У�
��3��������С���̵ķֶ�ֵΪ0.02A��ʾ��Ϊ0.3A����ʱ��ֵ����ĵ繦��P=UI=1.5��0.3A=0.45W��
��4���о����������Ĺ�ϵʱ��Ҫ���ֶ�ֵ�������˵ĵ�ѹ���䣬��5�����軻��10���ĵ�����ݷ�ѹԭ������ֵ����ֵõĵ�ѹ����Ҫ���ֶ�ֵ�������˵ĵ�ѹ���䣬������Ҫ��ȥ����ĵ�ѹ������Ӧ��������������·�еĵ��裬��Ƭ�����ƶ���ʹ��ѹ��ʾ��Ϊ1.5V��
��5������·�н���20������ʱ��Ҫ���ֵ�ѹ��ʾ��Ϊ1.5V���䣬������Ҫ��ȥ4.5V-1��5V=3V�ĵ�ѹ�����ݷ�ѹԭ��������������ֵ����Ϊ$\frac{3}{1.5}$��20��=40����Ϊ�������ṩ��4����ֵ������ʵ���ж������ϣ�Ӧѡ�á�50�� 1A���ı��裮
�ʴ�Ϊ����1������ͼ��ʾ��
��2����ֵ�����·��
��3��0.3�� 0.45��
��4���ң� 1.5��
��5����50�� 1A����
���� ����̽��ͨ����������뵼������ϵ�������·�����ӡ��������ϵķ����жϡ����ĵ�ѡ��ʵ��IJ������̡����Ʊ�������֪ʶ�����п����ȵ����⣮

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ��ľ�����ܼ⣬��������ѹǿ | |
| B�� | ˮ�ù����������������϶��������˴���ѹǿ | |
| C�� | ��ɽ�˶�Ա�ڵ�ɽ�����У��о����������ѣ�����Ϊ����ѹ��� | |
| D�� | �̶����ɻ������������Ϊ�����졢ѹǿ����������� |
| A�� | Ƶ�� | B�� | ���� | C�� | ��� | D�� | ��ɫ |
| ��Ʒ�ͺţ�KS-12 ���������1kg ���ѹ��220V�� �Ƶ�ʣ�50Hz ����ʣ�1100W ִ�б���----- |
��2����ˮ����������ʱ�ĵ������
��3����֪�õ���ˮ���ĵ�Դ�����ߵ���ԼΪ0.5��������ˮ�����У����Դ�����߷��ȶ��٣�
��4����ˮ����������5min���Ķ��ٵ��ܣ�
��5�����չ��ҹ涨�ı�������ˮ��ʹ�õĵ�Դ�����߲��ܹ��������������ܹ�С��Сǿ��ϸ�۲��˼����һЩ�����õ�������յ�����ů���ĵ�Դ��Ҳ���������ص㣬����һ�½��������涨��ԭ��
 ��ͼ��С���ݵĹ���ǡ�6V 3W������Դ��ѹΪ12V���պϿ���S������ƬP���е�ʱ������ǡ���������⣻�������¶ȵ�Ӱ�죬����ƬP�������ʱ��������
��ͼ��С���ݵĹ���ǡ�6V 3W������Դ��ѹΪ12V���պϿ���S������ƬP���е�ʱ������ǡ���������⣻�������¶ȵ�Ӱ�죬����ƬP�������ʱ��������| A�� | ���ݵĵ���Ϊ12�� | |
| B�� | ���ݵ�ʵ�ʵ���Ϊ0.5A | |
| C�� | �����������������ֵΪ24�� | |
| D�� | ��ƬP�������ʱ����·���ܹ���Ϊ4W |
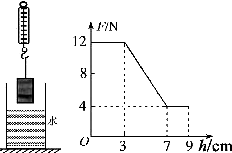 ��ͼ��ʾ�����ɲ����������һʵ��Բ���ݣ���Բ�����ʢ��ˮ�������Ϸ���ˮ��ijһ�߶ȴ������½��������ʼ����ˮ��ƽ�У���ʹ����û��ˮ��ijһ��ȴ�����ͼ�����������е��ɲ����Ƶ�ʾ��F��Բ�����½��߶�h�仯��ϵ������ͼ������
��ͼ��ʾ�����ɲ����������һʵ��Բ���ݣ���Բ�����ʢ��ˮ�������Ϸ���ˮ��ijһ�߶ȴ������½��������ʼ����ˮ��ƽ�У���ʹ����û��ˮ��ijһ��ȴ�����ͼ�����������е��ɲ����Ƶ�ʾ��F��Բ�����½��߶�h�仯��ϵ������ͼ������| A�� | Բ���������Ϊ12N | B�� | Բ�����ûʱ�ܵ��ĸ���Ϊ4N | ||
| C�� | Բ��������Ϊ4��10-4m3 | D�� | Բ������ܶ�Ϊ3��103kg/m3 |
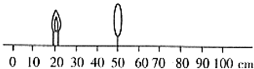 ��̽������������ɡ���ʵ���У��������Ľ�����10cm���ÿ̶ȳ���ˮƽ�����ϻ�һֱ�߲������̶ȣ������̶���50cm����
��̽������������ɡ���ʵ���У��������Ľ�����10cm���ÿ̶ȳ���ˮƽ�����ϻ�һֱ�߲������̶ȣ������̶���50cm����