��Ŀ����
2����̽�������ۻ�ʱ�¶ȱ仯�ص��ʵ���У���1������ͼ����ʾ��װ����װ��ʵ�����ģ�ʵ�������ѡ�ò�ͬ�Ĺ�����м��ȣ�ÿ��һ��ʱ������¶ȼƵ�ʾ���۲����״̬�ı仯��С���ñ���ʵ��ʱ�۲쵽���¶���ͼ����ʾ������������ȷ����B��ѡ�A������B����C������ʾ��Ϊ-12�森ͼ����С�λ��Ƶı����¶������ʱ��仯��ͼ��
��2������ͼ�۲쵽�����ó����ۻ�ʱ���ص��ǣ������������¶ȱ��ֲ���
��3�������������ۻ�ʱ�������ͬ�ص����CD
A������ B��ʯ�� C���� D������
��4�����һֱ���ձ����ȣ����ձ��е�ˮ����ʱ��С�Թ��е�ˮ�����ڣ�˵��ԭ���ᣬ��ΪС�Թ��е�ˮ�ܴﵽ�е㣬�����ܼ������ȣ�

���� ��1��ʹ���¶ȼƲ���Һ����¶ȣ�����ʱ����Ҫ��Һ����ƽ�������¶ȼƵķֶ�ֵ�����¶ȼ��е�Һ����ʲôλ�ã��Ӷ������¶�ֵ��
��2�����Ǿ��壬�ۻ����������ȡ��¶Ȳ��䣮
��3�������ľ����и��ֽ��������������������ķǾ�����ʯ�������������ࡢ���㣮
��4��Һ����ڵ��������ﵽ�е㣬�������ȣ�
��� �⣺��1��ʵ������У�ÿ��һ��ʱ������¶ȼƵ�ʾ���۲����״̬�ı仯��ʹ���¶ȼƲ���Һ����¶�ʱ������Ҫ��Һ����ƽ��B��������������¶ȼƵķֶ�ֵ��1�棬Һ������̶����£�ʾ��Ϊ-12�棻
��2�����Ǿ��壬�ۻ������������������¶ȱ��ֲ��䣮
��3���������������Ǿ��壻���ࡢ���㡢�������������ǷǾ��壮��ѡCD��
ˮ����ʱ���Թ������ˮ�����Դﵽ�е㣬�����Թ��ڵ�ˮҪ���Թ������ȣ����ߵ��¶���ͬ���Թ��ڵ�ˮ���ܴ��Թ���������������������Թ��ڵ�ˮ�ܴﵽ�е㣬���Dz��ܼ������ڣ�
�ʴ�Ϊ����1��״̬��B��-12����2�������������¶ȱ��ֲ��䣻��3��CD����4�����ᣬ��ΪС�Թ��е�ˮ�ܴﵽ�е㣬�����ܼ������ȣ�
���� ���⿼���¶ȼƵĶ�����������ۻ��ص㡢Һ����ڵ���������ͼ���ҳ���ص���Ϣ���۵㡢�ۻ������е��ص㣮ͬʱ�������˶��������ȶ��ٵķ�����

 ̽���빮�̺��Ͽ�ѧ����������ϵ�д�
̽���빮�̺��Ͽ�ѧ����������ϵ�д�| A�� | ̫������������� | |
| B�� | �����Dz����ٷֵ���С���� | |
| C�� | ���ӡ����Ӻ�ԭ���У��߶�������ԭ�� | |
| D�� | �������ѹ��˵��������Ӽ���϶ |
��1��������̥�Ե����ѹǿ�Ƕ��٣�
��2����������ι�·����ʻʱ��������Ϊ���ص�0.03�������ʱ���ڻ���ǣ���������Ĺ����ʷֱ��Ƕ��
��3�����������һ��ƽֱ�Ĺ�·��������ʻ8km�������͵�����m=4kg�����������������Ч�ʣ������͵���ֵq=4.5��107J/kg��
| ���� | 5��104N |
| ���ָ��� | 10 |
| ÿ����̥������� | 3��10-2m2 |
| �����ݻ� | 10m3 |
| A�� | �п��ڼ䣬ѧУ·�ν�ֹ�������ѣ������ڴ��������м������� | |
| B�� | ����������д������ٶ���340m/s | |
| C�� | ������װ�ĵ����״������û�����λ̽�����ϰ���� | |
| D�� | ���������Ƶ��Խ�ߣ����Խ�� |


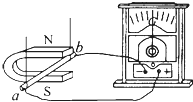 ��ͼ��ʾ���ѽ�����ab�ڴ����Ĵų��������˶�ʱ������������ͣ���м��ָ������ƫת�����ֲ�������������������ѧ�ϳ�Ϊ��Ÿ�Ӧ���������ֽ�����ab�����������������˶������Դ���Ϊ�����������ab��Դ��������ң�ѡ����ҡ������˶�����ʱ����������ͣ���м��ָ�������ѡ����ҡ���ƫת��
��ͼ��ʾ���ѽ�����ab�ڴ����Ĵų��������˶�ʱ������������ͣ���м��ָ������ƫת�����ֲ�������������������ѧ�ϳ�Ϊ��Ÿ�Ӧ���������ֽ�����ab�����������������˶������Դ���Ϊ�����������ab��Դ��������ң�ѡ����ҡ������˶�����ʱ����������ͣ���м��ָ�������ѡ����ҡ���ƫת�� ��ͼ��ʾ�ǽ����۵ij���ԭ��ͼ��ѡ����ӡ���Զ�ӡ����������۾�Ӧ��������������۾���
��ͼ��ʾ�ǽ����۵ij���ԭ��ͼ��ѡ����ӡ���Զ�ӡ����������۾�Ӧ��������������۾���