人类的生产生活离不开金属,下列三种金属使用的先后顺序依次为:铜、铁、铝。
(15) 金属被使用的先后顺序跟_____________(填序号)选项关系最大。
①地壳中金属元素的含量;②金属的导电性;③金属的活动性。
(16) 为探究锰与铁的金属活动性,在相同温度下,取大小相同、 表面光亮的两种金属薄片,分别投入等体积、等溶质的质量分数的足量稀盐酸中(反应中Mn显+2价),请回答:
表面光亮的两种金属薄片,分别投入等体积、等溶质的质量分数的足量稀盐酸中(反应中Mn显+2价),请回答:
| 金 属 | 铁 | 锰 |
| 与盐酸反应现象 | 放出气泡速率缓慢 | 放出气泡速率较快 |
| 结 论 | 活动性铁__________锰(填“>”“<”) |
除了比较与酸反应产生气体速率的快慢,请你再举出一种可比较这二种金属活动性强弱的方法(写出所需药品即可):_________________________________________。
(17) 将一定量的铝加入到硫酸亚铁和硫酸铜的混合溶液中,充分反应后过滤,向滤渣中加入盐酸,有气泡产生。则滤渣中一定有的物质是________________。(填化学式)
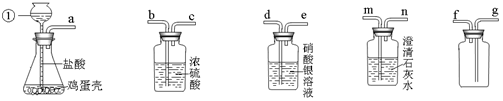

 ,在加热氯酸钾制取氧气的过程中,发现产生氧气的速率很慢,经检查不是KClO3变质,也不是装置气密性不好,你认为最可能的原因是 。
,在加热氯酸钾制取氧气的过程中,发现产生氧气的速率很慢,经检查不是KClO3变质,也不是装置气密性不好,你认为最可能的原因是 。
 根据右图A、B、C三种物质的溶解度曲线回答:
根据右图A、B、C三种物质的溶解度曲线回答: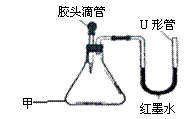 如右图是某同学设计的趣味实验装置,气密性良好。
如右图是某同学设计的趣味实验装置,气密性良好。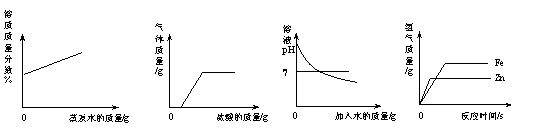 下列图①~④分
下列图①~④分