��Ŀ����
���û�ѧ�����ʾ�ӵ㲿�֣�����ʯ����Ҫ�ɷ�Ϊ
+4
| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
Al2O3
Al2O3
������Ԫ��| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
Fe2+
Fe2+
������ɫ���챦ʯ����Ҫ��ѧ�ɷ�Ҳ���������������ж�����Ԫ��������ѡ�| �� |
| ? |
Si
Si
���Ƶȣ�������Ҫ�Թ����ƣ�Na2SiO3������ʽ���ڣ���| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
| �� |
| ? |
| Ϊ |
| ? |
| �� |
| ? |
Na2
O3
| +4 |
| Si |
Na2
O3
���������й�Ԫ�غ���Ԫ�ص�����֮����| +4 |
| Si |
7��12
7��12
�����ȣ����������ݻ����ﻯѧʽ����д����������д���ɣ�
���ӵı�ʾ�������ڱ�ʾ�����ӵ�Ԫ�ط������Ͻǣ���������������������������������ǰ�����������ں�1�����ʱ��1Ҫʡ�ԣ�
����Ԫ�ط��ŵ���д����������д���ɣ�
����Ԫ�ػ��ϼ۵ı�ʾ������ȷ��������������Ҫ�����Ԫ�صĻ��ϼۣ�Ȼ�����仯ѧʽ��Ԫ�ص��Ϸ��������ź����ֱ�ʾ����������ǰ�������ں�����д��
���ݻ������и�Ԫ��������=��ԭ�ӵ����ԭ��������ԭ�Ӹ���֮�ȣ����з������
���ӵı�ʾ�������ڱ�ʾ�����ӵ�Ԫ�ط������Ͻǣ���������������������������������ǰ�����������ں�1�����ʱ��1Ҫʡ�ԣ�
����Ԫ�ط��ŵ���д����������д���ɣ�
����Ԫ�ػ��ϼ۵ı�ʾ������ȷ��������������Ҫ�����Ԫ�صĻ��ϼۣ�Ȼ�����仯ѧʽ��Ԫ�ص��Ϸ��������ź����ֱ�ʾ����������ǰ�������ں�����д��
���ݻ������и�Ԫ��������=��ԭ�ӵ����ԭ��������ԭ�Ӹ���֮�ȣ����з������
����⣺����������Ԫ����+3�ۣ���Ԫ����-2�ۣ��仯ѧʽΪAl2O3��
�����ӵı�ʾ�������ڱ�ʾ�����ӵ�Ԫ�ط������Ͻǣ���������������������������������ǰ�����������ں�1�����ʱ��1Ҫʡ�ԣ��������ӿɱ�ʾΪ��Fe2+��
��Ԫ�ص�Ԫ�ط���Ϊ��Si��
��Ԫ�ػ��ϼ۵ı�ʾ������ȷ��������������Ҫ�����Ԫ�صĻ��ϼۣ�Ȼ�����仯ѧʽ��Ԫ�ص��Ϸ��������ź����ֱ�ʾ����������ǰ�������ں������й�Ԫ�صĻ��ϼ�Ϊ+4�ۿɱ�ʾΪ��Na2
O3��
�������й�Ԫ�غ���Ԫ�ص�����֮��28����16��3��=7��12��
�ʴ�Ϊ��Al2O3��Fe2+��Si��Na2
O3��7��12��
�����ӵı�ʾ�������ڱ�ʾ�����ӵ�Ԫ�ط������Ͻǣ���������������������������������ǰ�����������ں�1�����ʱ��1Ҫʡ�ԣ��������ӿɱ�ʾΪ��Fe2+��
��Ԫ�ص�Ԫ�ط���Ϊ��Si��
��Ԫ�ػ��ϼ۵ı�ʾ������ȷ��������������Ҫ�����Ԫ�صĻ��ϼۣ�Ȼ�����仯ѧʽ��Ԫ�ص��Ϸ��������ź����ֱ�ʾ����������ǰ�������ں������й�Ԫ�صĻ��ϼ�Ϊ+4�ۿɱ�ʾΪ��Na2
| +4 |
| Si |
�������й�Ԫ�غ���Ԫ�ص�����֮��28����16��3��=7��12��
�ʴ�Ϊ��Al2O3��Fe2+��Si��Na2
| +4 |
| Si |
�����������ѶȲ�����Ҫ����ͬѧ�ǶԳ�����ѧ�����ѧʽ��Ԫ�ط��š����ϼۡ����ӷ��ŵȣ�����д������������

��ϰ��ϵ�д�
�����Ŀ
 )����ʽ���ڣ���������й�Ļ��ϼ۱�ʾΪ+4�� ���������й�Ԫ�غ���Ԫ�ص��������� �������ȣ�
)����ʽ���ڣ���������й�Ļ��ϼ۱�ʾΪ+4�� ���������й�Ԫ�غ���Ԫ�ص��������� �������ȣ� )����ʽ���ڣ���������й�Ļ��ϼ۱�ʾΪ+4�� ���������й�Ԫ�غ���Ԫ�ص��������� �������ȣ�
)����ʽ���ڣ���������й�Ļ��ϼ۱�ʾΪ+4�� ���������й�Ԫ�غ���Ԫ�ص��������� �������ȣ�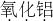 ________������Ԫ��
________������Ԫ��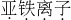 ________������ɫ���챦ʯ����Ҫ��ѧ�ɷ�Ҳ���������������ж�����Ԫ��������ѡ�
________������ɫ���챦ʯ����Ҫ��ѧ�ɷ�Ҳ���������������ж�����Ԫ��������ѡ�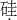 ________���Ƶȣ�������Ҫ�Թ����ƣ�Na2SiO3������ʽ���ڣ���
________���Ƶȣ�������Ҫ�Թ����ƣ�Na2SiO3������ʽ���ڣ���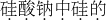

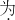 +4
+4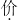 ________���������й�Ԫ�غ���Ԫ�ص�����֮����________�����ȣ�
________���������й�Ԫ�غ���Ԫ�ص�����֮����________�����ȣ� ______������Ԫ��
______������Ԫ�� ______������ɫ���챦ʯ����Ҫ��ѧ�ɷ�Ҳ���������������ж�����Ԫ��������ѡ�
______������ɫ���챦ʯ����Ҫ��ѧ�ɷ�Ҳ���������������ж�����Ԫ��������ѡ� ______���Ƶȣ�������Ҫ�Թ����ƣ�Na2SiO3������ʽ���ڣ���
______���Ƶȣ�������Ҫ�Թ����ƣ�Na2SiO3������ʽ���ڣ���

 +4
+4 ______���������й�Ԫ�غ���Ԫ�ص�����֮����______�����ȣ�
______���������й�Ԫ�غ���Ԫ�ص�����֮����______�����ȣ�