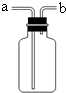
��Ŀ����
���������ʵ��װ��ͼ�ش����⡣
 |
(1)д������a��b�����ƣ�a�� ��b�� ��
(2)����Bװ����ȡ��������Ӧ�Ļ�ѧ����ʽ�� ��ѧ����
(3)ʵ�����ü����Ȼ�狀��������ƹ�������ķ�����ȡ������NH3����ͬʱ�õ��Ȼ��ƺ�ˮ���÷�Ӧ�Ļ�ѧ����ʽ�� ��Ӧѡ�õķ���װ��Ϊ ����װ�ñ�ţ���
(4)�ռ�����ʱӦѡ��Dװ�ã����ռ��������ļ���ƿ�����ڵ�����ɫ��̪��Һ��ˮ���У��۲쵽����ƿ���д�����ɫҺ����롣
����������Ϣ�ܽ�������������� ���ش��������ɣ���
![]() ��1���Թܣ���Һ©��
��1���Թܣ���Һ©��
![]() ��2��2H2O2 === 2H2O + O2��
��2��2H2O2 === 2H2O + O2��
��3��2NH4Cl + Ca(OH)2 === CaCl2 + 2NH3��+ 2H2O��A
��4���������ܶȱȿ���С������������ˮ��������ˮ��Һ�ʼ��Ե�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ