��Ŀ����
19�����ⶨijʯ��ʯ��Ʒ��̼��Ƶ�����������̼����ڸ����·ֽ����������ƺͶ�����̼��ʯ��ʯ��Ʒ�е��������Ȳ��ֽ⣬�Ҳ����ᷴӦ�����ס�����λͬѧ�ֱ��������������ʵ�鷽����������һ���ٳ�ȡʯ��ʯ��Ʒ����8g�����þƾ��Ƽ�����Ʒ��ֱ���������ٸı䣻�۽������������ڸ������������ȴ��Ƶ�����6.9g���ܼ��㣮
�����������ٳ�ȡʯ��ʯ��Ʒ����8g���ڼ�����������Ϊ7.3%������100g��ʹ̼�����ȫ��Ӧ������Ӧ�����Һ�м��뺬����3.2g������������Һ��ǡ���кͶ��������ܼ��㣮
��ش������й����⣺
��1������Ϊ���������У���ʵ���е��Ƿ���������ѡ����һ������ԭ����̼��Ʒֽ���Ҫ�ڸ��������½��У�
��2��100g��������Ϊ7.3%�������У������Ȼ��������Ϊ7.3g��
��3��������Ʒ��̼��Ƶ�����������
���� ���������£�̼��Ʒֽ����������ƺͶ�����̼��
̼��ƺ�ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼���������ƺ�ϡ���ᷴӦ�����Ȼ��ƺ�ˮ�������ṩ�����ݺͷ�Ӧ�Ļ�ѧ����ʽ���Խ�����ط���ļ��㣮
��� �⣺��1�����������У���ʵ���е��Ƿ���������ѡ����һ������ԭ����̼��Ʒֽ���Ҫ�ڸ��������½��У�
�������̼��Ʒֽ���Ҫ�ڸ��������½��У�
��2��100g��������Ϊ7.3%�������У������Ȼ��������Ϊ��100g��7.3%=7.3g��
���7.3��
��3����̼�������Ϊx����3.2g���������Ʒ�Ӧ���Ȼ�������Ϊy��
NaOH+HCl�TNaCl+H2O��
40 36.5
3.2g x
$\frac{40}{3.2g}$=$\frac{36.5}{y}$��
y=2.92g��
CaCO3+2HCl�TCaCl2+H2O+CO2����
100 73
x 7.3g-2.92g
$\frac{100}{x}$=$\frac{73}{7.3g-2.92g}$��
x=6g��
��������Ʒ��̼��Ƶ���������Ϊ��$\frac{6g}{8g}$��100%=75%��
�𣺷�������Ʒ��̼��Ƶ���������Ϊ75%��
���� ������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ�������ͬʱ�����˷������ݵ�����������ʱҪע��淶�Ժ�ȷ�ԣ�

| A�� | ���������ء�����Է�������Ϊ234 | |
| B�� | 1�����������ء������к���234������ | |
| C�� | 23.4�ˡ��������ء��к�����Ԫ�ص�����Ϊ8�� | |
| D�� | ���������ء���C��H��O����Ԫ�ص�������Ϊ12��10��5 |
| A�� | 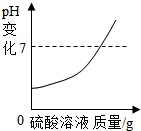 ������ˮ | B�� |  ������п | ||
| C�� |  ������NaOH��Һ | D�� |  ������BaCI2��Һ |
���й�������X�ɷֵķ����У���ȷ���ǣ�������
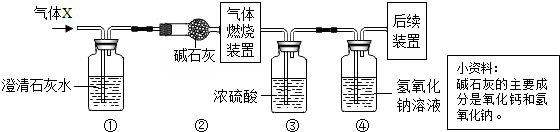
| A�� | һ���м��� | B�� | һ����H2 | C�� | ������CO2 | D�� | ���ܺ���H2��CO2 |
| A�� | 7.41 | B�� | 3.59 | C�� | 3.21 | D�� | 2.46 |
| A | B | C | D | |
| ���� ���� | Ӳˮ����ˮ | �����Ͷ�����̼ | ľ̿�ۺ�CuO��ĩ | �����������Ͷ������� |
| ����1 | �۲���ɫ | �ֱ�ͨ�����ʯ��ˮ���۲����� | ȡ�����ֱ����ϡ���ᣬ�۲����� | �۲���ɫ |
| ����2 | �ӵ����ķ���ˮ���۲����� | �ֱ���ȼ�ŵ�ľ�������뼯��ƿ�У��۲����� | �ֱ����ʯ���������գ��۲����� | �ֱ���뵽����������Һ�У��۲����� |
| A�� | A | B�� | B | C�� | C | D�� | D |
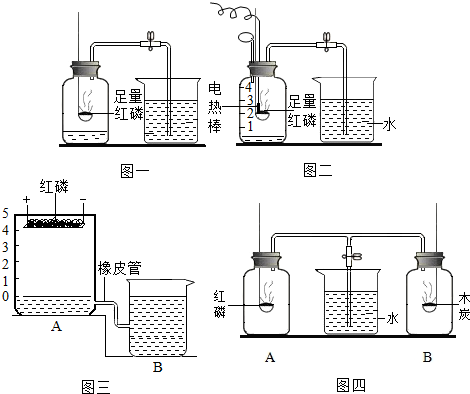
 ijʵ��С��ģ���¯�����Ļ�ѧ��Ӧԭ������ʵ�飬��װ����ͼ��ʾ����ش��������⣮
ijʵ��С��ģ���¯�����Ļ�ѧ��Ӧԭ������ʵ�飬��װ����ͼ��ʾ����ش��������⣮