��Ŀ����
��κ�������Ч����Լ��ʹ��ú��ʯ�͡���Ȼ����������������Ҫ�Ļ�ʯȼ�ϣ��ӽ��������������������Դ��ȫ�������ٵĹ�ͬ���⣬�Ƿ�չ��̼���õĵ���֮�����ش��������⣺
������1����Ȼ����̫ԭ�м�ͥ����Ҫ����ȼ�ϡ�Ϊ��ֹ
�� ��Ȼ��й©���Σ�գ����ڼ��а�װ���������ڼס���
��Ȼ��й©���Σ�գ����ڼ��а�װ���������ڼס���
��ͼ�У���װλ����ȷ���� ����ס����ҡ�����
д����Ȼ��ȼ�յĻ�ѧ����ʽ�� ��
������2����ֱ����ú������ȼ�ϼ���Ⱦ�������˷���Դ��úȼ�� ʱ������
ʱ������
���γ��������Ҫ�� ��֮һ��ú�������ǰ�ú��Ϊ�����Դ����Ҫһ��������һ����Ҫ��Ӧ�ǣ���ˮ����ͨ�����ȵ�ú�㣬�õ�һ����̼���������÷�Ӧ�Ļ�ѧ����ʽΪ ��
��֮һ��ú�������ǰ�ú��Ϊ�����Դ����Ҫһ��������һ����Ҫ��Ӧ�ǣ���ˮ����ͨ�����ȵ�ú�㣬�õ�һ����̼���������÷�Ӧ�Ļ�ѧ����ʽΪ ��
������3���ҹ������з���һ�ֵ綯����ʹ�õ���Դ������﮵�أ��ӻ�ѧ�Ƕȿ�����ʹ�����͵�����������ŵ���
�����ٳ����㼴�ɣ�
��1���� CH4+2O2 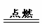 C
C O2+2H2O ��2��SO2����������� C+H2O====CO+H2
O2+2H2O ��2��SO2����������� C+H2O====CO+H2
��3������Ⱦ�����������̼�ŷŻ��Լ��ʯ��Դ�ȣ����������𰸾��ɣ�

 �п�������㾫��ϵ�д�
�п�������㾫��ϵ�д���ͬѧΪ�������������ƺ�����������Һ�����������ͼ��ʾ������ʵ�鷽����
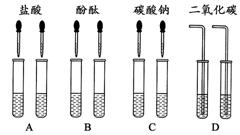
(1)�����ܴﵽʵ��Ŀ����__ __��
(2)��C��ʵ���з����Ļ�ѧ����ʽΪ__ _��
(3)ʵ�������ͬѧ��A��C����֧�Թ��е���Һ����ͬһ�����У�������Һ����ǣ�������˵����_(�ѧʽ)�������ɣ������̪����Һ�ʺ�ɫ��
(4)�����̪����Һ�ʺ�ɫ��˵����Һ�ʼ��ԡ�ʹ��Һ�ʼ��Ե�������ʲô�أ���ͬѧ��һ���Ʋ⣺��Һ�гʼ��Ե����ʿ�����̼���ơ��������ƺ�__ __���������е�һ�֣����������������������ɵĻ���
(5)Ϊ����֤�Ʋ⣬��ͬѧ�������ϣ���Ϥ�Ȼ�����Һ�����ԣ����������ʵ�������֤�������Ҫ��������пհף�
| ʵ������ | Ԥ������ | ���� |
| ȡ���������е��ϲ���Һ���Թ��У�����������Ȼ�����Һ������ | ||
| �����а�ɫ��������Һ����ɫ | ������̼���� | |
| �����������ɣ���Һ�ʺ�ɫ | �������������� | |
| �����а�ɫ��������Һ�ʺ�ɫ | _ _ |
(6)��ͬѧ��Ϊ��ͬѧ�ڢڲ���֤�Ľ��۲����ܣ�ԭ����_ __��
�����dz��л�ѧ������ʵ�飬��Ҫ��ش����⡣
 |
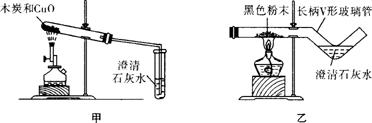
��1��ʵ��C���ձ��۵������� ��
��2���������ܴﵽʵ��Ŀ��ʵ���� ������ţ���Ϊʵ����Ӧʵ��Ŀ�ģ���ʵ��Ľ��ķ�����  ��
��
��3��ʵ��A��ʵ��ԭ���� ��
��ʵ��ⶨ���Ŀ����������ĺ���С�� ������ɵ�ԭ�������
������ɵ�ԭ�������
�� ����д�����㣩��

 C5H8O2 D��2KMnO4
C5H8O2 D��2KMnO4
 K2MnO4��MnO2��O2��
K2MnO4��MnO2��O2��