��Ŀ����
ijͬѧ��һƿ�治�ƶ����ֱ��ʵ�NaOH��������������̽��������Ϊ�����̽��ʵ�飺
��1��������⣺�������ɵ�������ʲô��
��2�����в��룺���ʺ������ɵ����ʿ������������������� ��д��ѧʽ����
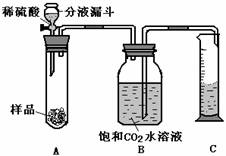
��3�����ʵ�飺���������ͼ��ʾ��һ��װ��������ʵ����֤��ͼ�е�������������ȥ����
��4��ʵ����֤��
�ٰ�ͼ���Ӻ�װ��
����������ƽȷ����2����Ʒ������A�Թ��У���B�е��뱥�͵�CO2��Һ��ƿ������
�۴ӷ�Һ©���м����Թ�����ϡ����رջ�������Ӧ������Ͳ���ռ�����Һ��Ϊ220ml��
a.�ж���Ʒ�ѱ��ʵ���������������������������������
b.�ڢٺ͢�֮��ȱ�ٵ�һ��ʵ�鲽��������������������������
c.B�еı���CO2��Һ������ˮ������������������������������������������������
d.�ж�A�е����ϡH2SO4�ѹ����ı�־����������������������������������������
e.����������Ʒ�б������ɵ����ʵ�����������(CO2�ܶ���1.8g/L����)������������ ��
��5���������ϡ������ܽ��������ȷ�ġ�
��6�����������ۣ�����װ���Dz���������ȷ�ⶨ�ѱ��ʵ���Ʒ��NaOH�����������ģ����������������������������������������� �������������������������� ��
��2��Na2CO3
��4��a��A�������������ɣ�ˮ������Ͳ
b�����װ�õ�������
c��CO2������ˮ����ˮ�����Ӱ��ʵ����
d�����������ݲ���
e��47.7%
��6��NaOH�������տ����е�ˮ�����ײ����������
