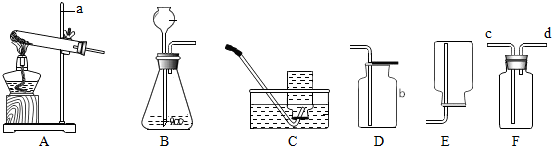
��Ŀ����
1����ѧ֪ʶ��������ϵ���У���ش���1������������Ʒ�ɽ��������Ƴɵ���D������ĸ����
A������ë�� B���������� C��ľ������ D�������ˮ��ͷ
��2��������ˮ�����¶���δ�ﵽ�е�ʱ��ˮ�г�������ð������˵��������ܽ�����¶ȵ����߶���С��
��3��С�ַ�������ϴ�õ�������ˮ�������ˣ������ԭ����������������е�������ˮ�ֵȹ�ͬ���õĽ��������ʱ�����������Ż�С���������Ϲ��ǣ����Ϩ���ˣ���ԭ���Ǹ���������
��4��С��������ʱ���ó涣ҧ�ˣ���ʹ�������ó���ڵ���������ԣ���Ӧ��ͿĨ�������ࣨ�ʳ�ᡱ�����ࡱ����
���� ��1�����ݽ������ϰ����������ͺϽ𣬾ݴ˽�ϳ������ʵ���ɽ��з����жϼ��ɣ�
��2������������ܽ�����¶ȵ����߶���С���
��3���������ڿ�������ʴ��ʵ���������������е�������ˮ��ͬ���õĽ�����������ԭ���������������ȼ��ڸ����������������ʹ�¶Ƚ�����ȼ����Ż�����£��ݴ˽��������з������
��4����������к͵�ԭ�����
��� �⣺
��1��A������ë�����������Ƴɵģ�������Ȼ���ϣ���ѡ�����
B���������������л��ϳɲ��ϣ���ѡ�����
C��ľ������������Ȼ���ϣ���ѡ�����
D���������ͷ���ò������ɵģ�����������ĺϽ����ڽ������ϣ���ѡ����ȷ��
��2��������ˮ�����¶���δ�ﵽ�е�ʱ��ˮ�г�������ð������˵��������ܽ�����¶ȵ����߶���С��
��3���������Ի���������Ϊ��������ˮ�ֳ�ֽӴ������������ͻȻ�Ż��ˣ��������Ϲ�������������˸������������ԭ����
��4������ʼ��ԣ�����кͣ������κ�ˮ���ó���ڵ���������ԣ���Ӧ��ͿĨ�������࣮
�𰸣���1��D����2����С����3��������ˮ�֣� ������������4�����࣮
���� �����ѶȲ����漰��֪ʶ��϶࣬��������Ӫ���ص����ࡢʳ����Դ����Ļ�ѧ��������ԭ��������ʴ���������ǽ����Ĺؼ���

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д� ��Уʵ������һƿ���ܲ������Լ������ȱ�ı�ǩ��ֻʣ�¡��ơ��͡�10%����������ͼ��ʾ������֪���dz��л�ѧ���õ��Լ���Сǿ��С��ͬѧ�ܸ���Ȥ����������ɷֽ���̽����
��Уʵ������һƿ���ܲ������Լ������ȱ�ı�ǩ��ֻʣ�¡��ơ��͡�10%����������ͼ��ʾ������֪���dz��л�ѧ���õ��Լ���Сǿ��С��ͬѧ�ܸ���Ȥ����������ɷֽ���̽����������⣺��ƿ�Լ�������ʲô��Һ�أ�
�������ۣ����������ǩ������жϣ���ƿ�Լ����������������������е�A������ţ���
A���� B���� C����
�������ϣ�
���л�ѧ�����ĺ��ƻ�������NaCl��NaOH��Na 2CO 3��NaHCO 3��
�ⶨ���£�20�棩ʱ���������ʵ��ܽ�ȵ����������
| ���� | NaCl | NaOH | Na 2CO 3 | NaHCO 3 |
| �ܽ��/g | 36 | 109 | 21.5 | 9.6 |
С�������Լ�ƿ��ע��������������10%���ϱ��е��ܽ�ȵ������жϣ���ƿ�Լ���������NaHCO3��
�������룺
�ٿ�����NaOH��Һ���ڿ�����Na2CO3��Һ���ۿ�����NaCl��Һ��
��Ʋ�ʵ�飺
��1��Сǿȡ��Ʒ�������μ���ɫ��̪��Һ��Һ����ɫ����Ϊ�������ȷ�������ж��Ƿ������ԭ���ǣ�
��2��С��Ϊ�˼������Һ������ʲô�ֽ���������ʵ�飺ȡ��Ʒ���Թ��У��μ��Ȼ�����Һ���а�ɫ�������ɣ����ó��������ȷ����Ӧ�Ļ�ѧ����ʽ��
| A�� | ��ɫ����ɫ����ɫ | B�� | ��ɫ����ɫ����ɫ | C�� | ��ɫ����ɫ����ɫ | D�� | ��ɫ����ɫ����ɫ |
| A�� | �� | B�� | �� | C�� | ̼ | D�� | �� |
| A�� | ��̼��������Ԫ����� | |
| B�� | ��̼��������Ԫ����� | |
| C�� | ��̼���⡢������Ԫ����� | |
| D�� | һ������̼��������Ԫ�أ����ܺ�����Ԫ�� |
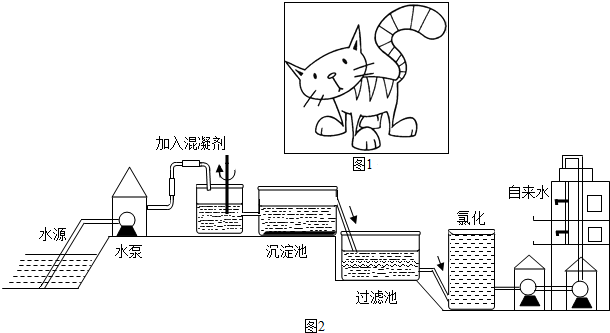
 ˮʱ����֮Դ��
ˮʱ����֮Դ��