��Ŀ����
����Ԩ���㣬�����˶���������ʫ�еġ��������ɣ�1��
�ϳ�
�ϳ�
�����Ƴɣ����㡱�������ṩ����ҪӪ�����ǣ�2��������
������
���������ǣ�3���ǹ�������ϸ���Ļ������ʣ��ǻ�����������������֯����Ҫԭ�ϲ��ṩ����
�ǹ�������ϸ���Ļ������ʣ��ǻ�����������������֯����Ҫԭ�ϲ��ṩ����
��������Ĺ����п��Լ����������ʹ�������������õĻ��ţ���������CaO2����ˮ��Ӧ�����������ƺ��������Ҫ�����壮�䷴Ӧ�Ļ�ѧ����ʽΪ��4��2CaO2+2H2O�T2Ca��OH��2+O2��
2CaO2+2H2O�T2Ca��OH��2+O2��
����Ӧǰ����Ԫ�صĻ��ϼ۷ֱ�Ϊ��5��-1�ۡ�-2�ۡ�-2�ۡ�0
-1�ۡ�-2�ۡ�-2�ۡ�0
����������1��ʫ�еġ��������������Ȳ����Ƴɵģ�
��2�������㸻����Ӫ���ؽ��з������
��3�����ݵ����ʵ��������ܽ��з������
��4����������CaO2����ˮ��Ӧ�����������ƺ��������Ҫ�����壨��������д����Ӧ�Ļ�ѧ����ʽ���ɣ�
��5�����ݵ�����Ԫ�صĻ��ϼ�Ϊ0���ڻ��������������ϼ۴�����Ϊ�㣬��Ϻ���Ԫ�صĻ�ѧʽ���н���⣮
��2�������㸻����Ӫ���ؽ��з������
��3�����ݵ����ʵ��������ܽ��з������
��4����������CaO2����ˮ��Ӧ�����������ƺ��������Ҫ�����壨��������д����Ӧ�Ļ�ѧ����ʽ���ɣ�
��5�����ݵ�����Ԫ�صĻ��ϼ�Ϊ0���ڻ��������������ϼ۴�����Ϊ�㣬��Ϻ���Ԫ�صĻ�ѧʽ���н���⣮
����⣺��1��ʫ�еġ��������������Ȳ����Ƴɵģ����������ںϳɲ��ϣ�
��2�������и��������ʣ�
��3���������ǹ�������ϸ���Ļ������ʣ��ǻ�����������������֯����Ҫԭ�ϲ��ṩ������
��4����������CaO2����ˮ��Ӧ�����������ƺ��������Ҫ�����壨����������Ӧ�Ļ�ѧ����ʽΪ��2CaO2+2H2O�T2Ca��OH��2+O2����
��5��CaO2�и�Ԫ����+2�ۣ���Ԫ�صĻ��ϼ�Ϊ-1�ۣ�H2O����Ԫ�صĻ��ϼ�Ϊ-2�ۣ�Ca��OH��2����Ԫ�صĻ��ϼ�Ϊ-2�ۣ��������ڵ��ʣ���Ԫ�صĻ��ϼ�Ϊ0��
�ʴ�Ϊ����1���ϳɣ���2�������ʣ���3���ǹ�������ϸ���Ļ������ʣ��ǻ�����������������֯����Ҫԭ�ϲ��ṩ��������4��2CaO2+2H2O�T2Ca��OH��2+O2������5��-1�ۡ�-2�ۡ�-2�ۡ�0��
��2�������и��������ʣ�
��3���������ǹ�������ϸ���Ļ������ʣ��ǻ�����������������֯����Ҫԭ�ϲ��ṩ������
��4����������CaO2����ˮ��Ӧ�����������ƺ��������Ҫ�����壨����������Ӧ�Ļ�ѧ����ʽΪ��2CaO2+2H2O�T2Ca��OH��2+O2����
��5��CaO2�и�Ԫ����+2�ۣ���Ԫ�صĻ��ϼ�Ϊ-1�ۣ�H2O����Ԫ�صĻ��ϼ�Ϊ-2�ۣ�Ca��OH��2����Ԫ�صĻ��ϼ�Ϊ-2�ۣ��������ڵ��ʣ���Ԫ�صĻ��ϼ�Ϊ0��
�ʴ�Ϊ����1���ϳɣ���2�������ʣ���3���ǹ�������ϸ���Ļ������ʣ��ǻ�����������������֯����Ҫԭ�ϲ��ṩ��������4��2CaO2+2H2O�T2Ca��OH��2+O2������5��-1�ۡ�-2�ۡ�-2�ۡ�0��
�����������ѶȲ������պϳɲ��ϵ������������ʵ�����������ʳ����Դ����ѧ����ʽ����д�����û��ϼ۵�ԭ�����ָ��Ԫ�صĻ��ϼ۵ķ�������ȷ�����Ĺؼ���

��ϰ��ϵ�д�
�����Ŀ
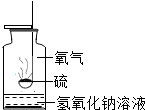 ����ͼװ������ȼ�յ�ʵ�飮
����ͼװ������ȼ�յ�ʵ�飮