��Ŀ����
8��ijѧϰС���������������о�����1������ϡ����������²�������Һ��pH���ߵ���ABC������ĸ����
A��������ˮ B������������
C���������������� D��������Ũ����
| ʱ��/h | 1 | 1.5 | 4 | 8 | 12 | 24 | 48 | 60 | |
| ��ˮ ����/g | ����� | 1.6 | 2.2 | 5.2 | 10.3 | 14.0 | 20.9 | 29.2 | 32.1 |
| ������ | 1.2 | 1.5 | 3.5 | 5.9 | 8.1 | 12.9 | 19.5 | 21.0 | |
������ȡ�����ʱ���Ӷ�����������������ȷ������£����������ҵ���������������ƫС���ƫ����ƫС�����䡱����
���ɱ���ʵ�����ݿ�֪���й�������ˮ�Ե�˵���������B������ĸ����
A��������Ҷ�����ˮ�� B��ֻ�����������ˮ�� C���������ˮ�Խ�������ǿ
��3��С��ͬѧ����ϡ�ͺ��ϡ��������ᱵ���������ơ�̼��ص����ʵ���Һ�ԡ����ֽⷴӦ������������������̽����
�ٽ�����������Һ������ϣ��ж�������֮���ܷ�����Ӧ����д�����з����кͷ�Ӧ�Ļ�ѧ����H2SO4+2NaOH�TNa2SO4+2H2O��
�����������������ʵ���Һ���ʵ����������ʻ�ϲ����ˣ��õ�һ����ɫ��Һ�����ⶨ������Һ�����ԣ����ڸ���Һ��һ�����е�������H+��Na+��K+��NO3-�����ܺ��е�������SO42-��
���� ��1��������Һ�������������֮��Ĺ�ϵ������
��2���ٸ��ݸ��Ӷ�������ȡ��Һ����������������ļ��㹫ʽ������
�ڸ��ݱ��е����ݷ����������ˮ�ԣ�
��3���ٸ������ʵ����ʣ�ѡ�����ʷֱ�д��һ���кͷ�Ӧ���г������ɵķ�Ӧ��
�ڸ�����Һ��Ϻ������ԣ�����������뱵���ӽ���γɳ�����������Ϻ����Һ��һ�����е����ӣ����ܺ��е����ӣ�
��� �⣺��1����Ϊ��ͬ�¶��£��������������Խ����Һ������Խǿ����Һ��pHԽС����ϡ�����м��������ˮʱ�����������������С����Һ��pH���û�з�����ѧ�仯����ϡ�����м�������ʱ���������ᷴӦ������ת��������Һ��pHҪ������ϡ�����м�����������ʱ���������غ����ᷴӦ����Һ��pHҪ�������ԣ���Һ��pH��ѧ�仯�����ߵ���ABC��
��2��������ȡʱŨ����ʱ�Ǹ��Ӷ�����ʵ����ȡ��Ũ�������ˣ������õ�ϡ�������������ƫС��
���ɱ���ʵ�����ݿ�֪��������Ҷ�����ˮ�ԣ��������ˮ�Խ�������ǿ����B����
��3�������������ʵ����ʿ�֪���������������Ʒ����кͷ�Ӧ����ѧ����ʽ�ǣ�H2SO4+2NaOH�TNa2SO4+2H2O��
��������Һ��Ϻ������ԣ����ԣ���Һ��Ӧ����H+�������� CO32-������������뱵���ӽ���γɳ�����һ�������ڱ����Ӻ���������ӣ����ԣ���Һ��һ�����е������У�H+��Na+��K+��NO3-����Һ�п��ܺ���SO42-��
�ʴ�Ϊ��
��1��ABC�� ��2����ƫС����B�� ��3����H2SO4+2NaOH�TNa2SO4+2H2O����H+��Na+��K+��NO3-��SO42-��
���� ������Ҫ�������й������֪ʶ��Ҫ�����������Ŀ����Ҫ������������Ļ�ѧ���ʡ���;����Ӧ����ͻ�ѧ����ʽ���Լ���֮��ص�֪ʶ��

 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д� �����ܿ����ϵ�д�
�����ܿ����ϵ�д�| A�� | 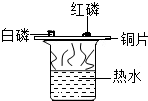 ̽����ȼ��ȼ�յ����� | B�� | 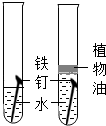 ̽������������� | ||
| C�� | 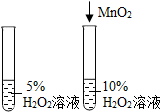 ̽���������̶Է�Ӧ���ʵ�Ӱ�� | D�� |  ̽�������ڲ�ͬ�ܼ��е��ܽ��� |
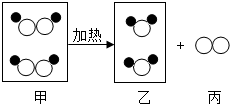 ��ͼ��ij��Ӧ����ʾ��ͼ������
��ͼ��ij��Ӧ����ʾ��ͼ������ ����ʾ��ԭ�ӣ�����ʾ��ԭ�ӣ������й�˵������ȷ���ǣ�������
����ʾ��ԭ�ӣ�����ʾ��ԭ�ӣ������й�˵������ȷ���ǣ�������| A�� | ͼ�мס��ҡ����������ʶ��������� | |
| B�� | �÷�Ӧ���ڷֽⷴӦ | |
| C�� | ͼ�мס��ҡ����������ʾ����ɷ��ӹ��ɵ� | |
| D�� | �÷�Ӧǰ��������ࡢԭ�������û�иı� |
 ������ȤС���ͬѧ�����ͼ��ʾ��ʵ�飬��������ʵ�鱨��
������ȤС���ͬѧ�����ͼ��ʾ��ʵ�飬��������ʵ�鱨�� ��10g CaCO3�в��ϼ���ϡ���ᣬ�������仯��ͼ��ʾ����ش��������⣺
��10g CaCO3�в��ϼ���ϡ���ᣬ�������仯��ͼ��ʾ����ش��������⣺ ����ͼ��������ˮ�����ϻش����⣮
����ͼ��������ˮ�����ϻش����⣮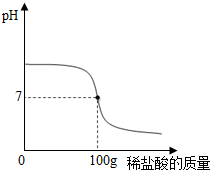 ���������׳���ʯ�ң����������������й㷺����;���������ѧ��ѧ֪ʶ�ش��������⣺
���������׳���ʯ�ң����������������й㷺����;���������ѧ��ѧ֪ʶ�ش��������⣺