��Ŀ����
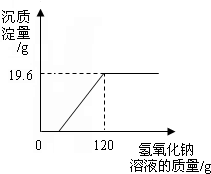
ij����С���һʪ��ұͭ���ŷŵķ�ˮ�к������������ͭ������Ⱦ�Ϊ�ⶨ�÷�ˮ�и���Ⱦ��ĺ�������ұͭ���ṩ������ˮ�IJο�������С���ͬѧ����������ʵ�飮ȡ��ˮ500g�������м���������������Ϊ20%������������Һ����ó��������������������������Һ��������ϵ��ͼ����ش��������⣺
��1��OA��û�в���������ԭ���ǣ��û�ѧ����ʽ��ʾ����______��
��2��C����Һ������Ϊ______��
��3���÷�ˮ������ͭ������������������д��������̣�

���𰸡���������1���������������ͭ����Һ�м�������������Һ��������������������з�Ӧ����������ȫ��Ӧʱ�������Ƽ���������ͭ������Ӧ����������ͭ��ɫ������
��2���ɷ�Ӧͼ���е����߿ɵ�֪B��ʱǡ����ȫ��Ӧ��C��ʱ�������ƹ�����Ȼ����ݷ�Ӧԭ��������Һ�е����ʣ�
��3��ǡ�÷�Ӧʱ���ɳ���19.6g��������������������ͭ��Ӧ�Ļ�ѧ����ʽ���������ɳ����������ɼ������ˮ������ͭ�����������������Һ�����ʵ�����������
����⣺��1���������������ͭ����Һ�м�������������Һ��������������������з�Ӧ����������ȫ��Ӧʱ�������Ƽ���������ͭ������Ӧ����������ͭ��ɫ������
����OA�����������������Ƶķ�Ӧ��
�ʴ�Ϊ��H2SO4+2NaOH=Na2SO4+2H2O��
��2��C��ʱ�������ƹ������������������Ʒ�Ӧ���������ƣ�����ͭ���������Ʒ�Ӧ����������ͭ�����������ƣ����Դ�ʱ��Һ�е������з�Ӧ���ɵ������ƺ������������ƣ�
�ʴ�Ϊ��Na2SO4��NaOH��
��3���⣺��500g��ˮ��CuSO4������Ϊ��
CuSO4+2NaOH=Cu��OH��2��+Na2SO4
160 98
�� 19.6g
 =
=
x=32g
���ˮ������ͭ����������Ϊ ×100%=6.4%
×100%=6.4%
��500g�÷�ˮ��CuSO4����������Ϊ6.4%��
���������⿼����ѧ����ͼ����ķ��������������ߵ�����ʾ���ᱻ��ȫ��Ӧ���۵��ʾ����ͭ����������ǡ����ȫ��Ӧ����ȷͼ���������ȷ���
��2���ɷ�Ӧͼ���е����߿ɵ�֪B��ʱǡ����ȫ��Ӧ��C��ʱ�������ƹ�����Ȼ����ݷ�Ӧԭ��������Һ�е����ʣ�
��3��ǡ�÷�Ӧʱ���ɳ���19.6g��������������������ͭ��Ӧ�Ļ�ѧ����ʽ���������ɳ����������ɼ������ˮ������ͭ�����������������Һ�����ʵ�����������
����⣺��1���������������ͭ����Һ�м�������������Һ��������������������з�Ӧ����������ȫ��Ӧʱ�������Ƽ���������ͭ������Ӧ����������ͭ��ɫ������
����OA�����������������Ƶķ�Ӧ��
�ʴ�Ϊ��H2SO4+2NaOH=Na2SO4+2H2O��
��2��C��ʱ�������ƹ������������������Ʒ�Ӧ���������ƣ�����ͭ���������Ʒ�Ӧ����������ͭ�����������ƣ����Դ�ʱ��Һ�е������з�Ӧ���ɵ������ƺ������������ƣ�
�ʴ�Ϊ��Na2SO4��NaOH��
��3���⣺��500g��ˮ��CuSO4������Ϊ��
CuSO4+2NaOH=Cu��OH��2��+Na2SO4
160 98
�� 19.6g
 =
=
x=32g
���ˮ������ͭ����������Ϊ
 ×100%=6.4%
×100%=6.4%��500g�÷�ˮ��CuSO4����������Ϊ6.4%��
���������⿼����ѧ����ͼ����ķ��������������ߵ�����ʾ���ᱻ��ȫ��Ӧ���۵��ʾ����ͭ����������ǡ����ȫ��Ӧ����ȷͼ���������ȷ���

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
 ij����С���һʪ��ұͭ���ŷŵķ�ˮ�к������������ͭ������Ⱦ�Ϊ�ⶨ�÷�ˮ�и���Ⱦ��ĺ�������ұͭ���ṩ������ˮ�IJο�������С���ͬѧ����������ʵ�飮ȡ��ˮ500g�������м���������������Ϊ20%������������Һ����ó��������������������������Һ��������ϵ��ͼ��
ij����С���һʪ��ұͭ���ŷŵķ�ˮ�к������������ͭ������Ⱦ�Ϊ�ⶨ�÷�ˮ�и���Ⱦ��ĺ�������ұͭ���ṩ������ˮ�IJο�������С���ͬѧ����������ʵ�飮ȡ��ˮ500g�������м���������������Ϊ20%������������Һ����ó��������������������������Һ��������ϵ��ͼ�� ��2012?̩����һģ��ij����С���һʪ��ұͭ���ŷŵķ�ˮ�к������������ͭ������Ⱦ�Ϊ�ⶨ�÷�ˮ�и���Ⱦ��ĺ�������ұͭ���ṩ������ˮ�IJο�������С���ͬѧ����������ʵ�飮ȡ��ˮ500g�������м���������������Ϊ20%������������Һ����ó��������������������������Һ��������ϵ��ͼ��
��2012?̩����һģ��ij����С���һʪ��ұͭ���ŷŵķ�ˮ�к������������ͭ������Ⱦ�Ϊ�ⶨ�÷�ˮ�и���Ⱦ��ĺ�������ұͭ���ṩ������ˮ�IJο�������С���ͬѧ����������ʵ�飮ȡ��ˮ500g�������м���������������Ϊ20%������������Һ����ó��������������������������Һ��������ϵ��ͼ��