��Ŀ����
13��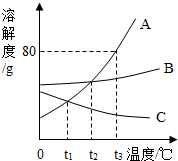 ��ͼ��A��B��C���ֹ������ʵ��ܽ������ͼ������ͼʾ�ش��������⣺
��ͼ��A��B��C���ֹ������ʵ��ܽ������ͼ������ͼʾ�ش��������⣺��1��t2��ʱ��A��B��C�������ʵ��ܽ�ȴ�С��ϵ��A=B��C��
��2��t3��ʱ����30gA���ʼ��뵽50gˮ�У�����ܽ��������Һ�Dz����� ������͡������͡�����Һ��������Һ������������Ϊ37.5%
��3�������£���ʢ��A�ı�����Һ���Թܷ���ʢˮ���ձ��У������ձ��ڵ�ˮ�м���һ�����IJ�NH4NO3�����裬�Թ��ڿ��Թ۲쵽���������о���������
��4����t2��ʱA��B��C���ֱ�����Һͬʱ������t1��ʱ��������Һ�����ʵ����������Ĵ�С��ϵ��B��A��C��
���� ���ݹ�����ܽ�����߿��ԣ��ٲ��ij������һ���¶��µ��ܽ�ȣ��Ӷ�ȷ�����ʵ��ܽ��ԣ��ڱȽϲ�ͬ������ͬһ�¶��µ��ܽ�ȴ�С���Ӷ��жϱ�����Һ�����ʵ����������Ĵ�С�����ж����ʵ��ܽ�����¶ȱ仯�ı仯������Ӷ��ж�ͨ�����½ᾧ���������ᾧ�ķ����ﵽ�ᴿ���ʵ�Ŀ�ģ�
��� �⣺��1��ͨ�������ܽ�����߿�֪��t2��ʱ��A��B��C�������ʵ��ܽ�ȴ�С��ϵ��A=B��C��
��2��t3��ʱ��A���ʵ��ܽ����80g�����Խ�30gA���ʼ��뵽50gˮ�У�����ܽ��������Һ�Dz�������Һ��������Һ������������Ϊ$\frac{30g}{80g}$��100%=37.5%��
��3��A���ʵ��ܽ�����¶ȵĽ��Ͷ���С�����������ˮ����Һ�¶Ƚ��ͣ����������£���ʢ��A�ı�����Һ���Թܷ���ʢˮ���ձ��У������ձ��ڵ�ˮ�м���һ�����IJ�NH4NO3�����裬�Թ��ڿ��Թ۲쵽���������о���������
��4��t1��ʱ��B���ʵ��ܽ�����A���ʴ�֮��A��B���ʵ��ܽ�����¶ȵĽ��Ͷ���С�����������壬C���ʵĽ����¶ȣ������������壬Ӧ�ð���t2��ʱ���м��㣬���Խ�t2��ʱ��A��B��C�������ʵı�����Һ������t1�棬������Һ����������������ϵ��B��A��C��
�ʴ�Ϊ����1��A=B��C��
��2�������ͣ�37.5%��
��3�����������
��4��B��A��C��
���� �����ѶȲ��Ǻܴ���Ҫ�����˹�����ܽ����������ʾ�����壬�����ݹ�����ܽ�������������ص����⣬�Ӷ������������⡢��������������

 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д�| A�� | 5%�Ĺ���������Һ | B�� | ����ȼ�պ�������� | ||
| C�� | ��ˮ����� | D�� | �ྻ�Ŀ��� |
| A�� |  �㵹Һ�� | B�� |  ���� | C�� |  ����Һ�� | D�� | 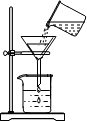 ���� |
| A�� | �����²��ij��Һ��pH=0�������Һһ�������� | |
| B�� | �������ĺ�ˮһ������ˮ | |
| C�� | �ܹ�������Ӧ������һ������ | |
| D�� | �μӷ�̪��Һ�Ժ�ɫ����Һһ���Ǽ����Һ |
| ѡ�� | ʵ��Ŀ�� | ʵ����� | ʵ������ | ʵ����� |
| A | ��֤ˮ����ɵ�ʵ�� | 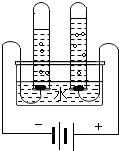 | �����������������������Ϊ2��1 | ˮ������Ԫ�غ���Ԫ����� |
| B | ��֤��ʯ�ұ��� | ȡ����ˮ�ܽ�������ɫ��̪��Һ | ��̪��� | ��ʯ���ѱ��ʣ������������� |
| C | ��ȥͭ�л��е�����ͭ | �ڿ��������� | ������ȫ��� | �õ�����������ͭ |
| D | ����NaOH��NH4NO3 | ��װ�е���ˮ���Թ��зֱ�������ֹ��壬���a��b | ��a���Թ�ˮ�����ߣ���b���Թ�ˮ�½��� | a��NaOH��b��NH4NO3 |
| A�� | A | B�� | B | C�� | C | D�� | D |
 ��ѧ��һ��ʵ���ѧ����ע���ʱ仯���̣�̽�����ʱ仯���������б�Ҫ��ij��ѧ��ȤС������ͼ��ʾ��ʵ��̽������ȼ�յ�������
��ѧ��һ��ʵ���ѧ����ע���ʱ仯���̣�̽�����ʱ仯���������б�Ҫ��ij��ѧ��ȤС������ͼ��ʾ��ʵ��̽������ȼ�յ�������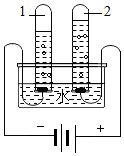 ˮ����������������ͨ�����������֮һ��
ˮ����������������ͨ�����������֮һ��