��Ŀ����
�ҹ��Ƽҵ������������°�̽�������ˡ������Ƽ���������������漰����Ҫ��ѧ��Ӧ���£�
��NH2��CO2��X��NH4HCO3[
��NH4HCO3��NaCl��NH4Cl��NaHCO3��
��2NaHCO3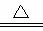 Na2CO3��H2O��CO2��
Na2CO3��H2O��CO2��
��ش��������⣺
��1����Ӧ����X�Ļ�ѧʽΪ��
��2����ȥ����Na2CO3��ĩ��������NaHCO3�ķ����� ��
��3����ҵ�����к����Ȼ��ƣ�ȡ55g��ҵ��������м���269.5gϡ���ᣬǡ����ȫ��Ӧ������22g������̼����
�ٹ�ҵ������̼���Ƶ�������������������������0.1%��
�ڷ�Ӧ����Һ�����ʵ�����������
H2O����������
�⣺���55g��ҵ������̼���Ƶ�����Ϊx���ڷ�Ӧ�������Ȼ��Ƶ�����Ϊy��
Na2CO3��2HCl=2NaCl��H2O��CO2��
106 117 44
x y 22g
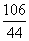 ��
��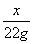 x��53g
x��53g
��ҵ������̼���Ƶ���������Ϊ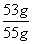 ��100%��96.4%
��100%��96.4%
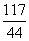 ��
��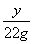 y��58.5g
y��58.5g
��Ӧ����Һ�������Ȼ��Ƶ���������Ϊ ��100%��20%
��100%��20%
�𣺢ٹ�ҵ������̼�����������Ϊ96.4%���ڷ�Ӧ����Һ�����ʵ���������Ϊ20%��

���й������漰��ѧ�仯���ǣ�������
| �� | A�� | �þ���ϩ�����Ƶ�ʳƷ�� | B�� | ��ʳ���Ƶô��� |
| �� | C�� | ��Һ̬���������Ƶ����� | D�� | ��ʯ�ͷ����Ƶ����� |
С����ʵ���ҷ���һƿδ֪Ũ�ȵ�����������Һ��Ϊ�����Ժ�ʹ�ã�������Ũ�Ƚ����˲ⶨ��ȡ20.0g������������Һ���ձ��У���εμ�������������Ϊ7.3%��ϡ���ᣬ����ʱ�Է�Ӧ�����Һ��pH�ƣ�һ�ֲⶨ��ҺpH���������ⶨ��Һ��pH���������������
| ����ϡ���� ������/g | 9.6 | 9.8 | 9.9 | 10.0 | 10.1 |
| ��Һ��pH | 12.4 | 12.1 | 11.8 | 7.0 | 2.2 |
�Իش�
��1�����μ�ϡ���������Ϊ9.8gʱ����Һ�е�����������������
��2��������������������Һ�����ʵ�����������

