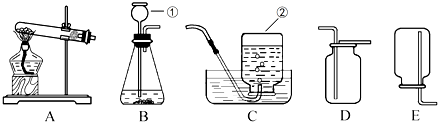
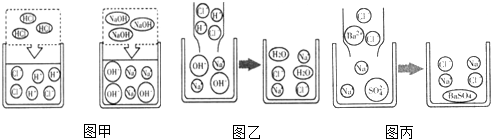
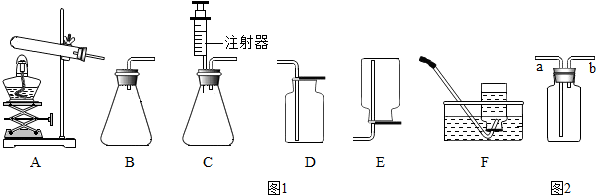
��Ŀ����
18��ij�о�ѧϰС�����ⶨʯ��ʯ��̼��Ƶ��������������õķ������£�ȡ��ʯ��ʯ��Ʒ16g����80gϡ������Ĵμ��룬ʵ��������������������֪ʯ��ʯ��Ʒ�к��еĶ�����������ʲ�����ˮ������ϡ���ᷴӦ��������ʵ�����ݼ��㣺| ʵ����� | 1 | 2 | 3 | 4 |
| ����ϡ���������/g | 20 | 20 | 20 | 20 |
| ʣ����������/g | 11 | 6 | 2.8 | n |
��2�������������ʵ�������������д��������̣�
���� ��1������ͼ�����ݿ�֪����1��2�η�Ӧ��������ʼ��ٵ�������Ϊ5g������3�η�Ӧ��������ʼ��ٵ�����Ϊ3.2g��˵����ʱ̼����ѷ�Ӧ�꣬���ٲ������壬�ʱ���n����ֵΪ2.8��
��2������ͼ�����ݿ�֪����1��2�η�Ӧ��������ʼ��ٵ�������Ϊ5g��˵��20gϡ����ǡ������ʯ��ʯ�е�5g̼�����ȫ��Ӧ������̼��������ᷴӦ�Ļ�ѧ����ʽ�͵�1����ȫ��Ӧ��̼��Ƶ����������ɼ������һ�β��뷴Ӧ��HCl������Ȼ��������ʵ�����������ʽ���㼴�ɣ�
��� �⣺��1������ͼ�����ݿ�֪����1��2�η�Ӧ��������ʼ��ٵ�������Ϊ5g������3�η�Ӧ��������ʼ��ٵ�����Ϊ3.2g��˵����ʱ̼����ѷ�Ӧ�꣬���ٲ������壬�ʱ���n����ֵΪ2.8��
�ʴ�Ϊ��2.8��
��2���������20gϡ����ǡ������ʯ��ʯ�е�5g̼�����ȫ��Ӧ��
���һ�β��뷴Ӧ��HCl����Ϊx��
CaCO3+2HCl=CaCl2+H2O+CO2��
100 73
5g x
$\frac{100}{73}=\frac{5g}{x}$����֮�ã�x=3.65g��
���������ʵ���������Ϊ��$\frac{3.65g}{20g}��100%$=18.25%��
�����������ʵ���������Ϊ18.25%��
�ʴ�Ϊ��
��1��2.8����2��18.25%��
���� ������Ҫ����ѧ�����û�ѧ����ʽ�����ʵ�����������ʽ���м��������������ס��¼�������ڷ����ű仯�����ݣ��������ݱ仯��ԭ���仯�Ĺ��ɣ����Ƿ���ʵ�����ݵ�һ�ֳ��÷�����


| A�� | �¶ȸ���t��ʱ��KCl���ܽ�ȴ���KNO3���ܽ�� | |
| B�� | ʹKCl����Һ�нᾧ���������ȴ�ȱ�����Һ�ķ��� | |
| C�� | 60���KNO3������Һ210g������40�棬������46g���� | |
| D�� | 40��ʱ��KNO3������Һ��������������Ϊ64% |
| A�� | ������γ� CO2+H2O�TH2CO3 | |
| B�� | ��ϡ����ϴȥ�������Թܱ��ϵ�ͭ Cu+H2SO4�TCuSO4+H2�� | |
| C�� | �ø������������ 2KMnO4$\frac{\underline{\;\;��\;\;}}{\;}$K2MnO2+MnO2+O2�� | |
| D�� | ����ʯ���γ�ԭ�� Ca��HCO3��2�TCaCO3��+H2O+CO2�� |