��Ŀ����
12��д�����з�Ӧ�Ļ�ѧ����ʽ������������ע��������Ӧ���ͣ���1�������ڿ�����ȼ�գ�4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5�����Ϸ�Ӧ��
��2����ʯ���кͻ�����Һ�е�ϡ����Ca��OH��2+H2SO4�TCaSO4+2H2O�����ֽⷴӦ��
��3���̬���ʲ�������Է��ϻ��ʹ�ã�����ɷ�Ч��ʧ��д���Ȼ������ʯ�ҷ�����Ӧ�Ļ�ѧ����ʽ2NH4Cl+Ca��OH��2�TCaCl2+2H2O+2NH3����
���� �����ڿ�����ȼ���������������ף�
�������ƺ�ϡ���ᷴӦ��������ƺ�ˮ��
�Ȼ�狀��������Ʒ�Ӧ�����Ȼ��ơ�ˮ�Ͱ�����
��� �⣺��1������ȼ�յĻ�ѧ����ʽΪ��4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5����Ӧ�������֣���������һ�֣����ڻ��Ϸ�Ӧ��
��2���������ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽΪ��Ca��OH��2+H2SO4�TCaSO4+2H2O�����ֻ�����������ɷ֣������������µĻ�������ڸ��ֽⷴӦ��
��3���Ȼ�狀��������Ʒ�Ӧ�Ļ�ѧ����ʽΪ��2NH4Cl+Ca��OH��2�TCaCl2+2H2O+2NH3����
���4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5�����Ϸ�Ӧ�� Ca��OH��2+H2SO4�TCaSO4+2H2O�����ֽ⣻ 2NH4Cl+Ca��OH��2�TCaCl2+2H2O+2NH3����
���� ��д��ѧ����ʽҪע���IJ���һ�Ƿ�Ӧ���������Ļ�ѧʽҪ��ȷ������Ҫ��ѭ�����غ㶨�ɣ�����ƽ������Ҫ�б�Ҫ�����������ǿ��Ƿ���Ҫ������������

��ϰ��ϵ�д�
�����Ŀ
2��ʹ���ᡢ��ȸ�ʴ��ҩƷʱ�����밴�ղ�����̽��У����д����������д�����ǣ�������
| A�� | �и�ʴ�Ե�ҩҺ�����۾��ڣ�Ҫ������ˮ��ϴ���в������������ | |
| B�� | ϡ��ɽ���Ƥ���ϣ������ô�����ˮ��ϴ����Ϳ��3%��5%��̼��������Һ | |
| C�� | ����ϡ����ʱ����������Ͳ�м���һ������Ũ���ᣬ������ע��ˮ�����Ͻ��� | |
| D�� | ����������Һ�����·��ϣ�����ˮ��ϴ����Ϳ��������Һ |
3��Ϊ�˶����Լ��Ķ���������С�������ڼ���ģ��ʵ�飬������С�����е�����ģ��ʵ����������в��ܴﵽʵ��Ŀ���ǣ�������
| A�� | ��ʳ��ˮȥ������е�ˮ�� | B�� | �ÿ���������ʵ�� | ||
| C�� | ��ʳ���������� | D�� | �������ᡢ��ɰ�ǡ�С�մ�������ˮ |
20��С��������ͬѧ��ʵ���ҷ�����һ��ʵ�����������ƿ��������������Һ���Լ�ƿƿ����Χ��һЩ��ɫ���壮���ǻ�������������Һ�Ѿ����ʣ����ǽ���������ʵ��̽����
��������⡿����������Һ�Ƿ���ʣ�
��������롿
�ٸ�����������Һû�б��ʣ��ڸ�����������Һ���ֱ��ʣ��۸�����������Һȫ�����ʣ�
��ʵ����֤��
�����ۡ�С��ͬѧ��Ϊ����������ʵ�飨1����2�����ƶϣ��������ȷ����С������Ϊ�����Ҳ������ȷ��������̼������ҺҲ��ʹ��̪��Һ��죮
����С������������²���ʵ�飺
��1��ȡԭ��Һ�������Թ��У���μ���BaCl2��Һ�����ٲ������������ã��ֲ㣮
��2��ȡ�ϲ���Һ�������м���CuSO4��Һ��������ɫ������������˵������������Һ���ֱ��ʣ���ѧ����ʽ��CuSO4+2NaOH�TNa2SO4+Cu��OH��2������֮��˵������������Һ�Ѿ�ȫ�����ʣ�
����˼��������������Һ���ʵ�ԭ������ǣ��û�ѧ����ʽ��ʾ����2NaOH+CO2�TNa2CO3+H2O��
����չ���졿ȡ���ֱ��ʵ�����������Ʒ10g�μ�������ϡ�������2.2g�Ķ�����̼������Ʒ��̼���Ƶ�����������
��������⡿����������Һ�Ƿ���ʣ�
��������롿
�ٸ�����������Һû�б��ʣ��ڸ�����������Һ���ֱ��ʣ��۸�����������Һȫ�����ʣ�
��ʵ����֤��
| ���� | ���� | ���� | ��ѧ����ʽ | |
| ��1�� | ȡ������Һ���Թ��У��μ���ɫ��̪��Һ | ��ɫ��̪��� | ��Һ�Լ��� | / |
| ��2�� | ȡ������Һ���Թ��У��μ�ϡ���� | �����ݲ��� | ��Һ�д���̼������ӣ���̼���ƣ� | Na2CO3+2HCl�T2NaCl+H2O+CO2���� |
����С������������²���ʵ�飺
��1��ȡԭ��Һ�������Թ��У���μ���BaCl2��Һ�����ٲ������������ã��ֲ㣮
��2��ȡ�ϲ���Һ�������м���CuSO4��Һ��������ɫ������������˵������������Һ���ֱ��ʣ���ѧ����ʽ��CuSO4+2NaOH�TNa2SO4+Cu��OH��2������֮��˵������������Һ�Ѿ�ȫ�����ʣ�
����˼��������������Һ���ʵ�ԭ������ǣ��û�ѧ����ʽ��ʾ����2NaOH+CO2�TNa2CO3+H2O��
����չ���졿ȡ���ֱ��ʵ�����������Ʒ10g�μ�������ϡ�������2.2g�Ķ�����̼������Ʒ��̼���Ƶ�����������
7����ͼ��ij�ܱ����������ʱ仯����ʾ��ͼ�����У��� ��������ԭ�ӣ���
��������ԭ�ӣ��� ��������ԭ�ӣ�����˵������ȷ���ǣ�������
��������ԭ�ӣ�����˵������ȷ���ǣ�������
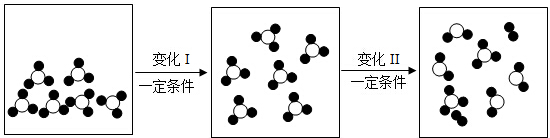
 ��������ԭ�ӣ���
��������ԭ�ӣ��� ��������ԭ�ӣ�����˵������ȷ���ǣ�������
��������ԭ�ӣ�����˵������ȷ���ǣ������� 
| A�� | �仯I�еġ�һ�������������������¶� | |
| B�� | �仯�������ǷֽⷴӦ | |
| C�� | �仯���з�Ӧ�漰�����ַ���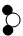 �� �� �� �� �ĸ�����Ϊ2��1��2 �ĸ�����Ϊ2��1��2 | |
| D�� | ��ʾ��ͼ���������ʾ�Ϊ������ |
17��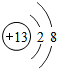 ��ͼΪijԪ�ص�һ�����ṹʾ��ͼ������˵������ȷ���ǣ�������
��ͼΪijԪ�ص�һ�����ṹʾ��ͼ������˵������ȷ���ǣ�������
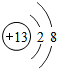 ��ͼΪijԪ�ص�һ�����ṹʾ��ͼ������˵������ȷ���ǣ�������
��ͼΪijԪ�ص�һ�����ṹʾ��ͼ������˵������ȷ���ǣ�������| A�� | ��Ԫ����һ�ֽ���Ԫ�� | B�� | ��Ԫ��ԭ�Ӻ�����2�����Ӳ� | ||
| C�� | ������������ | D�� | ���������ȶ��ṹ |
1����ƽ���˵��ձ��ж�ʢ��100g7.3%�����ᣬ��ƽƽ�⣬�����10gþ�����Ļ����ұ���ag̼��ƣ���ַ�Ӧ����ƽƽ�⣬��a����ֵΪ��������
| A�� | 9.8g | B�� | 10g | C�� | 14.2g | D�� | 17.9g |
4���������ʵ���;���������ʵ������������������仯ѧ���ʵ��ǣ�������
| A�� | ��ī��д���� | B�� | ʯī���缫 | C�� | �ɱ�������� | D�� | ��̿ұ������ |
