��Ŀ����
16�� ij��ѧС��Ϊ�ⶨһƿʧȥ��ǩ����������������������ֳɼס������飬�ֱ��ò�ͬ�ķ������вⶨ��
ij��ѧС��Ϊ�ⶨһƿʧȥ��ǩ����������������������ֳɼס������飬�ֱ��ò�ͬ�ķ������вⶨ��ʵ����ƣ�
���飺ȡ����̼��Ʒ�ĩ5g���ձ��У�����20g��������Һ����ַ�Ӧ���ˡ�ϴ�ӡ����������ʣ�����3g��
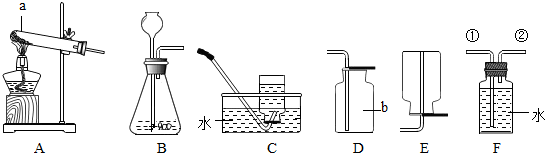
���飺ȡ����̼��Ʒ�ĩ5g����ƿ�У���20g��������Һ�����Һ©�������òⶨ����������̼�������20�桢������ѹ����������Ũ�ȣ�װ��ʾ��ͼ��ͼ��
���ݴ�����
��1�������ü������ݼ�������������������������д��������̣�
��2�������������С�ڼ��飮�������ʵ���Եõ���ͬ�����������Ͳ������������ܵ�ԭ����
�ۢܣ���������ţ���
�ٲ�����ˮ���������������������
��B��C֮��ĵ�����ʵ��ǰ��ˮ��ʵ�����ˮ
��������ѹ��ͬʱ��������̼������Ӧ�����ˮѹ����Ͳ
�ܶ�����̼�����ܽ���A�з�Ӧ�����Һ�У�
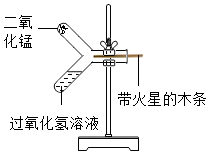
���� ��1������ʵ���ԭ����̼��ƺ����ᷴӦ��ͨ������̼����������������������
��2��������̼��ƺ����ᷴӦ���������壬�����������������ϡ�����������������������IJ����������ܵ��¶ȡ��ܽ�ȵ��������Ӱ�죬�������е�ͼ���������ڵμ�����ʱ������Ľ��뼴ʹ����Ӧ��Ҳ�ᵼ��B�е�ˮ����C�������ʹ������ƫ�ߣ�����ĿҪ���о����ƫ�ͣ���Ȼ��û�����ÿ������������Ӱ�죬����˼��ʱҪ������ĿҪ����У�
��� �⣺
��1������̼��Ʒ�Ӧ����5g-3g=2g
��ϡ�������������Ϊx
CaCO3+2HCl=CaCl2+H2O+CO2��
100 73
2g 20gx
$\frac{100}{73}$=$\frac{2g}{20gx}$
x=7.3%
�𣺸������������������7.3%��
��2�������ԭ�����ö�����̼��������������������������ȼ��������̼������Ϊm������ϡ�������������x������������Һ������Ϊn��
��ϡ�������������Ϊx
CaCO3+2HCl=CaCl2+H2O+CO2��
73 44
nx m
$\frac{73}{44}$=$\frac{nx}{m}$
x=$\frac{73m}{44n}$ ���Կ���m������ƫСʱ���ᵼ���������������ƫС��
�ٲ�����ˮ�������������������������ᵼ���������������ƫ��
��B��C֮��ĵ�����ʵ��ǰ��ˮ��ʵ�����ˮ�������ڵ�ˮ�������˶�����̼��������ˮ��Ҳ�͵��¶�����̼������ƫ��Ҳ�͵����������������ƫ��
��������ѹ��ͬʱ��������̼������Ӧ�����ˮѹ����Ͳ��ѹ����ˮƫ�٣����Ӧ�Ķ�����̼ƫС��������̼������ȡ�����Ҳ�ı�������ˮ�������Ǿ�����ν�����˶��ٶ�����̼��Ҳ����ˮ�������Ӧ������̼������������ϡ�������������ƫС��
�ܶ�����̼�����ܽ���A�з�Ӧ�����Һ�У����ֶ�����̼��Aװ�����գ������B������ƫ�٣����Ӧ��ˮҲ��ƫС�����յ��������������ƫС��
��ѡ���ۢܣ�
���� ������Ҫ��ʵ����Ƶ�ԭ�����з�������ԭ���ĽǶȷ������ֿ��ܵ�Ӱ��Ҫ�أ�

| A�� | ����������ʢ������ͭ��Һ��ԭ��Fe+CuSO4�TFeSO4+Cu �û���Ӧ | |
| B�� | ����ʴ��ԭ��4Al+3O2�TAl2O3 ������Ӧ | |
| C�� | ��ҵ���Գ�����Ϊԭ��ұ��������2Fe2O3+3C $\frac{\underline{\;����\;}}{\;}$4Fe+3CO2�� ��ԭ��Ӧ | |
| D�� | ���ˮ����������������2H2O $\frac{\underline{\;���\;}}{\;}$2H2��+O2�� �ֽⷴӦ |
 ��ѧ��ʦ�ں�ͬѧ��̽���������̵Ĵ�����ʱ���Խ̲�ʵ������˴�����ƣ�
��ѧ��ʦ�ں�ͬѧ��̽���������̵Ĵ�����ʱ���Խ̲�ʵ������˴�����ƣ�
 �����£����ס��ҡ������ֹ���ֱ�������֧�Թܺ�һ���ձ���ˮ�У�������ͼ1��ʾ���ס���������ʣ�࣬�ҹ���ȫ���ܽ⣮���µ�80��ʱ�����ּ���ȫ���ܽ⣬�ҹ���������������û�����Ա仯��
�����£����ס��ҡ������ֹ���ֱ�������֧�Թܺ�һ���ձ���ˮ�У�������ͼ1��ʾ���ס���������ʣ�࣬�ҹ���ȫ���ܽ⣮���µ�80��ʱ�����ּ���ȫ���ܽ⣬�ҹ���������������û�����Ա仯��