��Ŀ����
ʯӢ����Ҫ�ɷ�Ϊ��������(SiO2)�����Dz�����ҵ���մɹ�ҵ��ԭ�ϣ�ұ��ҵ�����ۼ���
(1)��������Ľṹ����ʯ���ƣ�������________(����ӡ���ԭ�ӡ������ӡ�)���ɵģ���������________(����ڡ������ڡ�)�����
(2)װ������������Һ���Լ�ƿ�����ò�������ԭ�����ڳ����£�NaOH�벣�����е�SiO2�����ط�����Ӧ����Na2SiO3��H2O��Na2SiO3ʹƿ����ƿ������һ����÷�Ӧ�Ļ�ѧ����ʽΪ��________________________________________________��
(8��)(1)ԭ�ӡ�����
(2)2NaOH��SiO2===Na2SiO3��H2O

��ϰ��ϵ�д�
 ���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д�
���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д� ����ѵ��ϵ�д�
����ѵ��ϵ�д�
�����Ŀ
�Ƚϡ�������������ѧϰ��ѧ����Ҫ������ͨ���ȽϿ��Է�����ͬ�ķ�Ӧ���ڲ�ͬ��������»ᷢ����ͬ�ķ�Ӧ�������ʾ�����ɷ�Ӧ���ɣ�
| ��� | ��ѧ����ʽ | ���� |
| ʾ�� | C��O2(����) C��O2(������) | ��Ӧ�� ��ͬʱ����Ӧ�������ͬ�������ﲻͬ |
| (1) | 4Na��O2===2Na2O 2Na��O2 | |
| (2) | 3Cu��8HNO3(ϡ)===3Cu(NO3)2��2NO����4H2O Cu��4HNO3(Ũ)===Cu(NO3)2��2NO2����2H2O |
 4CO2��2H2O������Ȳ�Ļ�ѧʽ��(����)��
4CO2��2H2O������Ȳ�Ļ�ѧʽ��(����)��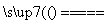 2Fe2O3
2Fe2O3 2KCl��3O2��
2KCl��3O2��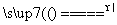 2P2O5
2P2O5