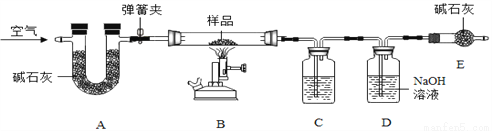
��Ŀ����
ij��ѧ��ȤС��ͬѧ�Լ�����ͨ�ɵ���к��е����ʲ�����Ũ�����Ȥ�����ǰ������ʱ�ŵ���ζ�ҷ����к�ɫ���ʡ�
��������⣩Ϊʲô�а�ζ����ɫ���ʵijɷ���ʲô��
��������裩���ŵ���ζ˵���������(��NH4Cl)����ɫ������������ۡ�����ͭ��������̡�
��ʵ��̽����ͬѧ�Ƿֱ����ʵ�����̽����
ʵ��I��С��ȡ������ɫ������______________��������в�����ĥ���ᵽǿ�Ұ�ζ������ʪ��ĺ�ɫʯ����ֽ��⣬�۲쵽��ֽ������֤����ɫ�����к�����Ρ�
ʵ��II��С������ɫ���ʼ���������ˮ�н����ܽ⣬���ú�______��ȡ���������ʵ�顣
ʵ�鲽�� | ʵ������ | ʵ����� |
1.ȡ������ɫ����װ���Թ��У��ټ������ϡ���� | ���������ɣ���Һ����ɫ | ________________ |
2��ȡ������ɫ����װ����һ֧�Թ��У��ټ���______��Һ���������Թ�����������ǵ�Сľ���� | _________________ | ��ɫ��ĩ�к��ж������� |
���������ۣ�����2�з�����Ӧ�Ļ�ѧ����ʽΪ______________��
��ʯ�� ���� ��ɫ��ĩ��һ����������������ͭ H2O2 �����������ݣ�ľ����ȼ 2H2O22H2O��O2�� ��������[ʵ��̽��]ʵ����������ϻ�ų�����������ȡ������ɫ��������ʯ�һ�������в�����ĥ���ᵽ��ζ������ʪ���ʯ����ֽ����Ƿ����ɰ�������ֽ������֤����ɫ�����к�����Σ�ʵ���С������ɫ���ʼ���������ˮ�н����ܽ⣬���õ���������Ըò������ǹ��ˣ�1.ȡ������ɫ����װ���Թ�...
��ϰ��ϵ�д�
 ���㼤�������100�ִ��Ծ�ϵ�д�
���㼤�������100�ִ��Ծ�ϵ�д�
�����Ŀ


 B.
B.  C.
C.  D.
D.