��Ŀ����
1��������С��ͬѧ����100g10%NaCl��Һ��ʵ�����ʾ��ͼ��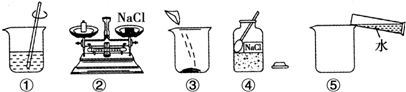
��1����ʵ����ȷ�IJ���˳���Ǣܢڢۢݢ٣�����ţ�
��2�����ܽ�Ĺ����в���������������Ǽ����ܽ�
��3������۲�ָ��ͼ�д���IJ�������ڣ�����ţ�
��4�����Ƹ���Һ��Ҫ�Ȼ���10g����Ҫˮ90mL��ˮ���ܶ�Ϊ1g/mL��
��5��С��ͬѧ��������ϵ�ʳ��ת�Ƶ��ձ�ʱ������������ʳ�������������ϣ�������ʹ������Һ��������������С�ڣ�����ڡ������ڡ���С�ڡ���10%��
���� ��1��������һ������������������Һ�IJ���Ϊ�����㡢��������ȡ���ܽ�������
��2�����ܽ�Ĺ����в���������������Ǽ����ܽ⣻
��3����������ƽ����ҩƷ������ʱ��Ҫ������������̣�ҩƷ�������̣�
��4���������ʵ�����=��Һ�����������ʵ�������������Ȼ��Ƶ��������������ˮ�������������
��5���Ӳ���������ʳ�������������ϣ�ʹ�ܽ���Ȼ��Ƶ��������ٷ������
��� �⣺��1������һ������������������Һ�IJ���Ϊ�����㡢��������ȡ���ܽ⣬������ȷ��˳��Ϊ���ܢڢۢݢ٣�
��2�����ܽ�Ĺ����в���������������Ǽ����ܽ⣻
��3����������ƽ����ҩƷ������ʱ��Ҫ������������̣�ҩƷ�������̣���ͼ����������룬������ҩƷ�����Դ���
��4���������ʵ�����=��Һ�����������ʵ����������ɵã���Ҫ�Ȼ��Ƶ�����Ϊ��100g��10%=10g��ˮ������Ϊ��100g-10g=90g��90g��1g/mL=90mL��
��5������������ʳ�������������ϣ�ʹ�ܽ���Ȼ��Ƶ��������٣��ܼ����������䣬��ʹ���ʵ�����������С��
�ʴ�Ϊ����1���ܢڢۢݢ٣���2�������ܽ⣻
��3���ڣ�
��4��10��90��
��5��С�ڣ�
���� ������Ҫ��������Һ�����Ʋ��衢ע�����������ƽ��ʹ�÷��������ʵ��������������֪ʶ��ƽʱע����й��ɺ��ܽᣮ

 ������������ϵ�д�
������������ϵ�д�| A�� | 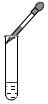 | B�� | 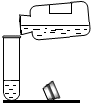 | C�� | 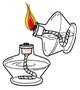 | D�� | 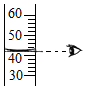 |
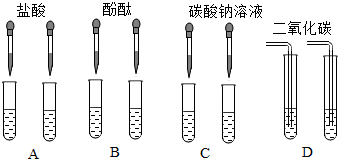
��1�������ܴﵽʵ��Ŀ��ʵ�鷽����CD��������ĸ��
��2��д������ʵ�鷽�����г������ɵķ�Ӧ����ʽ��Na2CO3+Ca��OH��2�TCaCO3��+2NaOH��Ca��OH��2+CO2�TCaCO3��+H2O��
��3��ʵ�������ͬѧ��A��C����֧�Թ��е���Һ����ͬһ�����У�������Һ����ǣ������̪����Һ�ʺ�ɫ����ͬѧ�ж���Һ�ʼ��ԣ������룺ʹ��Һ�ʼ��Ե����ʿ�����̼���ơ��������ƺ����������е�һ�֣������������������������ɵĻ���
��4��Ϊ����֤���룬��ͬѧ�������ϣ���Ϥ�Ȼ�����Һ�����ԣ����������ʵ�������֤�������Ҫ��������пհף�
| ʵ������ | Ԥ������ | ���� |
| ȡ���������� ���ϲ���Һ�� �Թ��У����� �������Ȼ��� ��Һ������ | �����а�ɫ��������Һ����ɫ | �ʼ��Ե������� |
| �����������ɣ���Һ�ʺ�ɫ | �ʼ��Ե��������������� | |
| �����а�ɫ��������Һ�ʺ�ɫ | �ʼ��Ե�������̼���ƺ��������� |
��������⡿���ձ�����Һ�е�������ʲô��
��������롿����ͬѧ�Է�Ӧ����Һ��������������ֲ��룬�����ٲ���һ�ֲ��룺
����һ������ΪNa2SO4��
�����������ΪNa2SO4��NaOH��
������������ΪNa2SO4��H2SO4��NaOH��
�����ģ���1������ΪNa2SO4��H2SO4��
��2��ͨ��������С��ͬѧ��Ϊ���������������������������ƺ�����ܹ��森
���������ϡ�Na2SO4��Һ������
��ʵ��̽����
����ͬѧ�������ʵ����̣�����д�±���
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ��3��ȡ��Ӧ���ձ��е���Һ���Թܣ������̪��Һ | ���������ȷ |
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ��4�� | ��������ȷ |
��5���ձ��е���Һĩ������ֱ�ӵ�����ˮ����������ɵ�Σ������ȾˮԴ����ʴ��ˮ���ȣ�дһ������
��6���ڷ�����ѧ��Ӧ���������ʳɷ�ʱ���������������⣬���迼�Ƿ�Ӧ���Ƿ������
[����A]��[����B]��[����C]��[����A]��
| A�� | Fe��Fe2O3��FeSO4��Fe | B�� | CO��CO2��H2CO3��CO | ||
| C�� | NaOH��NaCl��Na2CO3��NaOH | D�� | HCl��CuCl2��BaCl2��HCl |
| A�� | CaCl2 | B�� | CaCl2 H2O | ||
| C�� | CaCl2 Ca��ClO��2 | D�� | CaCl2 Ca��ClO��2H2O |
| A�� | ����þ���� | B�� | �ơ�þ���� | C�� | þ�������� | D�� | ���ж� |