��Ŀ����
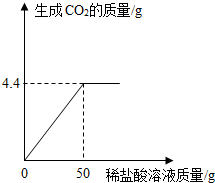 ij��ѧ��ȤС��Ϊ�ⶨ���Ȼ��ƺ�̼������ɵĻ������̼���Ƶ���������������������ʵ�飺ȡ100g�������ȫ�ܽ�������ˮ�У�����μ���ϡ������ɵ�CO2���������ϡ����ķ�Ӧ��ϵ��ͼ��ʾ��������CO2����ȫ��������������ܽ���ˮ�������������㣺����������������С�����һλ����
ij��ѧ��ȤС��Ϊ�ⶨ���Ȼ��ƺ�̼������ɵĻ������̼���Ƶ���������������������ʵ�飺ȡ100g�������ȫ�ܽ�������ˮ�У�����μ���ϡ������ɵ�CO2���������ϡ����ķ�Ӧ��ϵ��ͼ��ʾ��������CO2����ȫ��������������ܽ���ˮ�������������㣺����������������С�����һλ����
��1����100�˻������̼������ռ����������Ϊ���٣�
��2������ϡ��������ʵ���������Ϊ���٣�
�⣺��������̼���Ƶ�����Ϊx������������HCl������Ϊy
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 73 44
x y 4.4g
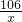 =
=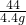 x=10.6g
x=10.6g
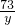 =
=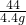 y=7.3g
y=7.3g
��100�˻������̼������ռ����������=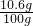 ��100%=10.6%
��100%=10.6%
����ϡ��������ʵ���������= ��100%=14.6%
��100%=14.6%
�𣺣�1����100�˻������̼������ռ����������Ϊ10.6%����2������ϡ��������ʵ���������Ϊ14.6%��
��������1����100�˻������̼������ռ����������= ��100%��������ͼ�ɵ÷�Ӧ���ɶ�����̼���������ɶ�����̼�������ݷ�Ӧ�Ļ�ѧ����ʽ��̼���Ƶ������õ������
��100%��������ͼ�ɵ÷�Ӧ���ɶ�����̼���������ɶ�����̼�������ݷ�Ӧ�Ļ�ѧ����ʽ��̼���Ƶ������õ������
��2������ϡ��������ʵ���������=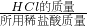 ��100%������ϡ��������������ͼ�ɵã�HCl���������ɶ�����̼���������ݷ�Ӧ�Ļ�ѧ����ʽ������ã�
��100%������ϡ��������������ͼ�ɵã�HCl���������ɶ�����̼���������ݷ�Ӧ�Ļ�ѧ����ʽ������ã�
���������ν���������б仯�����ߵ��۵��ʾ��Ӧǡ����ȫ��Ӧʱ��������ʵ�������ϵ��
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 73 44
x y 4.4g
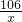 =
=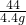 x=10.6g
x=10.6g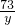 =
=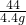 y=7.3g
y=7.3g��100�˻������̼������ռ����������=
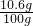 ��100%=10.6%
��100%=10.6%����ϡ��������ʵ���������=
 ��100%=14.6%
��100%=14.6%�𣺣�1����100�˻������̼������ռ����������Ϊ10.6%����2������ϡ��������ʵ���������Ϊ14.6%��
��������1����100�˻������̼������ռ����������=
 ��100%��������ͼ�ɵ÷�Ӧ���ɶ�����̼���������ɶ�����̼�������ݷ�Ӧ�Ļ�ѧ����ʽ��̼���Ƶ������õ������
��100%��������ͼ�ɵ÷�Ӧ���ɶ�����̼���������ɶ�����̼�������ݷ�Ӧ�Ļ�ѧ����ʽ��̼���Ƶ������õ��������2������ϡ��������ʵ���������=
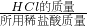 ��100%������ϡ��������������ͼ�ɵã�HCl���������ɶ�����̼���������ݷ�Ӧ�Ļ�ѧ����ʽ������ã�
��100%������ϡ��������������ͼ�ɵã�HCl���������ɶ�����̼���������ݷ�Ӧ�Ļ�ѧ����ʽ������ã����������ν���������б仯�����ߵ��۵��ʾ��Ӧǡ����ȫ��Ӧʱ��������ʵ�������ϵ��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
 ij��ѧ��ȤС��Ϊ�ⶨʵ������һƿ���õĹ���������Һ�����ʵ�������������������ʵ�飮
ij��ѧ��ȤС��Ϊ�ⶨʵ������һƿ���õĹ���������Һ�����ʵ�������������������ʵ�飮 ��2012?��ɽ��ģ��ij��ѧ��ȤС��Ϊ�ⶨһ��ʯ��ʯ��Ʒ��̼��Ƶ�������������������ʵ�飺�ٳ�ȡ25.0gʯ�ҿ���Ʒ��ƽ���ֳ����ݣ����ֱ�ӹ��ɿ�״�ͷ�ĩ״�� �ڽ�������Ʒ�ֱ�Ͷ��������ƿ�У���������������ͬ��������������ϡ���ᣨ��Ʒ�������ʲ�����ˮҲ�����ᷴӦ����������ɶ�����̼�������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ��
��2012?��ɽ��ģ��ij��ѧ��ȤС��Ϊ�ⶨһ��ʯ��ʯ��Ʒ��̼��Ƶ�������������������ʵ�飺�ٳ�ȡ25.0gʯ�ҿ���Ʒ��ƽ���ֳ����ݣ����ֱ�ӹ��ɿ�״�ͷ�ĩ״�� �ڽ�������Ʒ�ֱ�Ͷ��������ƿ�У���������������ͬ��������������ϡ���ᣨ��Ʒ�������ʲ�����ˮҲ�����ᷴӦ����������ɶ�����̼�������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ��