��Ŀ����
(6��)ʵ������һ�Ȼ��ƺ��Ȼ�����ɵ���Ʒ��Ϊ�ⶨ�����Ȼ��ƵĴ��Ȳ����Ƴ�һ������������������Һ��ij��ѧ̽��С��ȡһ�������ĸ���Ʒ��������ʵ�顣
��ش��������⣺

��1��д����Ӧ�ڵĻ�ѧ����ʽ ��
��2��������֪�����г������вμӷ�Ӧ�Ĵ�������(X)�ı���ʽ
��3������Ʒ���Ȼ��Ƶ���������Ϊ ��
��4������36.5%��Ũ������������ǡ����ȫ��Ӧ��ϡ���ᣬ���ˮ�����Ϊ mL��
��5�����������õ�c��ҺΪֻ��һ�����ʵIJ�������Һ����c��Һ�����ʵ���������Ϊ ��
��6������ҵ����58.5t�������Ȼ��Ƶ���ƷΪԭ������ȡ�������ƣ����Ƶ��������Ƶ�����Ϊ ��
��1��Na2CO3+ 2HCl =2 NaCl+ CO2��+H2O����2��= ����3��80%�� ��4��80����5��22.6%����6��32t �������� �����������μӷ�Ӧ��̼���Ƶ�����Ϊx �Ȼ���ΪY�����ɵ��Ȼ��Ƶ�����ΪZ CaCl2+Na2CO3===2NaCl+CaCO3�� 111 106 117 100 Y x z 10�� �μӷ�Ӧ�Ĵ������� (X...
��ϰ��ϵ�д�
�����Ŀ
�����������ʵ�������ӵ�֪ʶ������ȷ���ǣ� ��
ѡ�� | ��ʵ | ���� |
A | Ũ���᳨�ڷ���һ��ʱ����������������С | ˮ���Ӳ����˶���Ũ���� |
B | ̼�����������ֽ��̼�������Ȳ��ֽ� | ��ͬ�����ʵķ������ʲ�ͬ |
C | ˮ�տ���ˮ�������ױ����� | �¶����߷����˶��ӿ죬������ |
D | �л�����Ŀ�����쳣�Ӵ� | ԭ�ӵ����з�ʽ��ͬ |
A. A B. B C. C D. D
D ��������A��Ũ���᳨�ڷ���һ��ʱ����������������С������ΪŨ������лӷ��ԣ�Ũ�����е��Ȼ�������˶���������ȥ�ˣ�����B��̼�����������ֽ��̼�������Ȳ��ֽ⣬̼�����ƺ�̼���ƾ��������ӹ��ɵģ����ǵ������Ӿ�Ϊ�����ӣ������ӷֱ���̼��������ӡ�̼������ӣ��������ǵ������Ӳ�ͬ�����ʲ�ͬ������C��ˮ�տ���ˮ�������ױ���������Ϊ�¶����߷����˶��ӿ죬���Ӽ�ļ���������������...



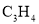 O)���ж����ʣ��������彡���������йر�ϩȩ��˵������ȷ����
O)���ж����ʣ��������彡���������йر�ϩȩ��˵������ȷ����