��Ŀ����
 ���ж�����ɽ��ʢ��ʯ��ʯ��ijУ����С��Ϊ�˽�ʯ��ʯ��Դ��Ʒ�ʣ��������ַ����Ե���ʯ�Ľ��л�ѧ������
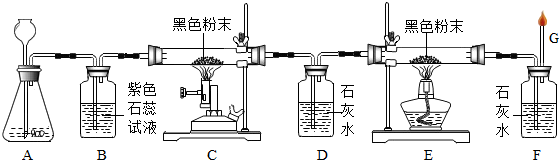
���ж�����ɽ��ʢ��ʯ��ʯ��ijУ����С��Ϊ�˽�ʯ��ʯ��Դ��Ʒ�ʣ��������ַ����Ե���ʯ�Ľ��л�ѧ����������һ��ȡ10gʯ��ʯ��ĩ��Ʒ������ͼ��ʾװ�ã���ּ����������㶨����ȴ��Ƶù�������Ϊ8.9g��
����������ȡ10gͬ�ʵ�ʯ��ʯ������Ʒ�������ձ��м�����ϡ���ᣬ��ַ�Ӧ��Ƶû�����������������4.27g��
������ϣ�����ʯ��ʯ���������ʵĻ�ѧ���ʾ����ȶ������Ȳ��ֽ��Ҳ������ᷢ����ѧ��Ӧ��
����������ݣ���������ַ�����õ�ʯ��ʯ��̼��Ƶ�������������ȷ��0.1%����
��1������һ��̼��Ƶ�����������
��2����������̼��Ƶ����������Ƕ��٣���д�����ⲽ�裩
��3���Ƚ�����ʵ�鷽���������������ϴ��ԭ�������ʲô��
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
��������1��̼������ȷֽ����������ƺͶ�����̼��������ٵ������������ɶ�����̼�����������ݻ�ѧ����ʽ��ͨ��������̼�����������̼��Ƶ�������Ȼ�����ʯ��ʯ��̼��Ƶ�����������
��2��̼��ƺ����ᷴӦ���ɶ�����̼���壬��Ӧ����������������ٵ������������ɶ�����̼�����������ݶ�����̼�ɼ���̼��Ƶ���������������̼��Ƶ�������������̼��Ʒֽ���Ҫ���·������ַ��������ԭ��
��2��̼��ƺ����ᷴӦ���ɶ�����̼���壬��Ӧ����������������ٵ������������ɶ�����̼�����������ݶ�����̼�ɼ���̼��Ƶ���������������̼��Ƶ�������������̼��Ʒֽ���Ҫ���·������ַ��������ԭ��
����⣺��1�����ɶ�����̼������Ϊ10g-8.9g=1.1g��������1.1g������̼��̼��Ƶ�����Ϊx��
CaCO3
CaO+CO2��
100 44
x 1.1g
=
x=2.5g
ʯ��ʯ��Ʒ��̼��Ƶ�����������
��100%=25%
������������������4.27g˵��������4.27g������̼������Ʒ�е�̼��Ƶ�����Ϊy��
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
y 4.27g
=
y=9.7g
ʯ��ʯ��Ʒ��̼��Ƶ�����������
��100%=97%��
ʯ��ʯ�ֽ���Ҫ���£����������ϴ��ԭ������Ǿƾ��ƻ����¶�ƫ�ͣ�ʯ��ʯ��ĩδ�ܳ�ַֽ⣬�������ϴ�
�ʴ�Ϊ��
��1��42.7%��
��2��97.0%��
��3�������Ǿƾ��ƻ����¶�ƫ�ͣ�ʯ��ʯ��ĩδ�ܳ�ַֽ⣬�������ϴ�
CaCO3
| ||
100 44
x 1.1g
| 100 |
| 44 |
| x |
| 1.1g |
x=2.5g
ʯ��ʯ��Ʒ��̼��Ƶ�����������
| 2.5g |
| 10g |
������������������4.27g˵��������4.27g������̼������Ʒ�е�̼��Ƶ�����Ϊy��
CaCO3+2HCl�TCaCl2+H2O+CO2��
100 44
y 4.27g
| 100 |
| 44 |
| y |
| 4.27g |
y=9.7g
ʯ��ʯ��Ʒ��̼��Ƶ�����������
| 9.7g |
| 10g |
ʯ��ʯ�ֽ���Ҫ���£����������ϴ��ԭ������Ǿƾ��ƻ����¶�ƫ�ͣ�ʯ��ʯ��ĩδ�ܳ�ַֽ⣬�������ϴ�
�ʴ�Ϊ��
��1��42.7%��
��2��97.0%��
��3�������Ǿƾ��ƻ����¶�ƫ�ͣ�ʯ��ʯ��ĩδ�ܳ�ַֽ⣬�������ϴ�
������������Ҫ����ѧ������ȫ��Ӧ�ĸ�������⣬�Լ����û�ѧ����ʽ������������ʽ���м�������������ݻ�ѧ����ʽ����ʱһ��Ҫע���ʽ�ͼ����ȷ�ԣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
����ʵ�鲻�ܴﵽʵ��Ŀ���ǣ�������
A�� ����CO2���� |
B�� �����������Ͻ�Ƥ�� |
C�� Ϩ��ƾ��� |
D�� Һ����� |
2012��12��22�գ��Ϻ�������ؿ������¶���-������Դ��������������в���������Դ���ǣ�������
| A��̫���� | B������ | C��ʯ�� | D������ |


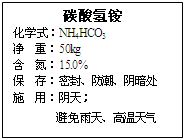 ��ͼ�����ֻ��ʱ�ǩ��һ���֣���ش�
��ͼ�����ֻ��ʱ�ǩ��һ���֣���ش�