��Ŀ����
2011��8 ��23�յ�26����˻������ڱ�Ļ�����ʵ�ʢ���������������̵�ӡ��
��1�����˻���ʹ�ñ��飨C3H8����ȼ�ϣ�������______��Ԫ����ɣ���Ԫ�ص�������Ϊ______������һ��______��ѡ��л�����������
��2�����˻��ڼ䣬���ֹ��������ö����ѣ�C2H6O����ȼ�ϣ���������ȫȼ�����ɶ�����̼��ˮ�Ļ�ѧ����ʽ��______��
��3��̫���ܵ�����Դ�ڴ˴δ��˻��еõ��㷺��Ӧ�ã�������һ������Դ�����ƣ�______��
�⣺��1����Ϊ����Ļ�ѧʽ��C3H8�����Ը������к�������Ԫ�أ�����̼������Ԫ�ص�������Ϊ��12��3��1��8=9��2�����������ں�̼�Ļ��������л��
��2���÷�Ӧ�ķ�Ӧ���Ƕ����Ѻ������������Ƕ�����̼��ˮ���䷽��ʽΪ��C2H6O+3O2 2CO2+3H2O��
2CO2+3H2O��
��3������������Դ�з��ܡ�ˮ�ܡ���ϫ�͵��ȵȣ�
�ʴ�Ϊ������1������Ԫ�أ�9��2���л��
��2��C2H6O+3O2 2CO2+3H2O��
2CO2+3H2O��
��3�����ܣ�ˮ�ܡ���ϫ�͵��ȵȺ������ɣ���
��������1�����ݱ���Ļ�ѧʽ�������㲢�ж����������༴�ɣ�
��2�����ݻ�ѧ����ʽ����дҪ����д���ɣ�
��3��������֪������Դ��֪ʶ�شɣ�
�����������ǶԻ�ѧʽ������Ŀ��飬��������˻�ѧ��Դ�����������Ҳ�����������������������������صĻ�ѧ֪ʶ����ѧϰ������Ҫ������ϵʵ�ʣ���ʵ�������֪ʶ��������ѧ֪ʶȥ���ʵ�����⣮
��2���÷�Ӧ�ķ�Ӧ���Ƕ����Ѻ������������Ƕ�����̼��ˮ���䷽��ʽΪ��C2H6O+3O2
 2CO2+3H2O��
2CO2+3H2O����3������������Դ�з��ܡ�ˮ�ܡ���ϫ�͵��ȵȣ�
�ʴ�Ϊ������1������Ԫ�أ�9��2���л��
��2��C2H6O+3O2
 2CO2+3H2O��
2CO2+3H2O����3�����ܣ�ˮ�ܡ���ϫ�͵��ȵȺ������ɣ���
��������1�����ݱ���Ļ�ѧʽ�������㲢�ж����������༴�ɣ�
��2�����ݻ�ѧ����ʽ����дҪ����д���ɣ�
��3��������֪������Դ��֪ʶ�شɣ�
�����������ǶԻ�ѧʽ������Ŀ��飬��������˻�ѧ��Դ�����������Ҳ�����������������������������صĻ�ѧ֪ʶ����ѧϰ������Ҫ������ϵʵ�ʣ���ʵ�������֪ʶ��������ѧ֪ʶȥ���ʵ�����⣮

��ϰ��ϵ�д�
 ��������ܸ�ϰϵ�д�
��������ܸ�ϰϵ�д�
�����Ŀ
 Si+4R���÷�Ӧ��R�Ļ�ѧʽ��________��
Si+4R���÷�Ӧ��R�Ļ�ѧʽ��________��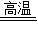 Si+4R���÷�Ӧ��R�Ļ�ѧʽ�� ��
Si+4R���÷�Ӧ��R�Ļ�ѧʽ�� ��