题目内容
向20g锌样品(杂质不溶于水,也不溶于酸)中加入100g质量分数为19.6%的稀硫酸,恰好完全反应。试计算该锌样品中锌的质量分数(写出计算过程)。
解:设20g锌样品中含有锌的质量为x ································································· 1分
Zn + H2SO4 == ZnSO4 + H2↑ ································································ 1分
65 98
x 100g×19.6%
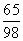 =
=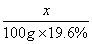 ······························································ 2分
······························································ 2分
x =13g ······························································ 1分
锌样品中锌的质量分数=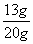 ×100%=65% ····················································· 1分
×100%=65% ····················································· 1分
答: 锌样品中锌的质量分数为65%。

 各地期末复习特训卷系列答案
各地期末复习特训卷系列答案 小博士期末闯关100分系列答案
小博士期末闯关100分系列答案下列四个图像中能正确表示对应变化关系的是
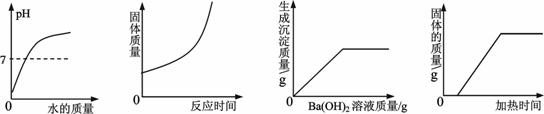
| |
A B C D
A.向一定量稀盐酸中加水稀释 B.高温煅烧一定质量的石灰石
C.向一定量的稀硫酸中加入Ba(OH)2溶液 D.加热一定质量的碳和氧化铜的混和物
某同学在油菜地不慎被蜜蜂氮蜇伤,蜜蜂的刺液呈酸性,为了减轻痛痒,在蜇伤处涂抹的物质是( )
|
| A. | 牛奶(pH≈6) | B. | 苹果汁(pH≈3) | C. | 牙膏(pH≈9) | D. | 矿泉水(pH≈7) |
下图是今年我市中考化学实验操作考查中,某些学生的实验操作示意图,其中正确的是( )
|
| A. |
取用锌粒 | B. |
过滤泥水 | C. |
加热铜片 | D. |
检验氧气 |
下列说法中正确的是( )
①餐具上的油污可用加入洗涤剂的水洗掉,是因为洗涤剂具有乳化作用
②将氧气压缩后储存于钢瓶中,是因为氧分子变小了
③盐酸能够导电,是因为HCl在水中会解离出H+和Cl﹣
④氢气可用于冶炼金属,是因为氢气具有可燃性.
|
| A. | ①③ | B. | ②③ | C. | ②④ | D. | ③④ |
实验室可用于氯酸钾抽取氧气的装置组合是( )
|
| A. | ①④ | B. | ②③ | C. | ①⑤ | D. | ②⑤ |