��Ŀ����
ij����ұ���������������������⣬�����ɷֲ�����ˮ��Ҳ�����ᷴӦ�������������ⶨ����������������������������ش��������⣺��1������250g������������Ϊl 9.6%��ϡ���ᣬ��Ҫ98%��Ũ�����������______g��
��2������������飬�������ܽ�Ļ�ѧ����ʽΪ______��
��3����17g��������ǡ����150g��1 9.6%��ϡ������ȫ��Ӧ����
�����17g��������������������x���ı���ʽΪ______��
�ڴ�������������������������Ϊ______��
����������Һ�м���39.8�˵�ˮ������������Һ�����ʵ���������Ϊ______��
��4��������������������Ҫ���Ͻ�108�֣�����95%���������������У�����ʧ��5%������Ҫ�����������������______�֣�
���𰸡���������1��ϡ��ǰ�����ʵ��������䣮
��2���������������ᷴӦ�����κ�ˮд����ѧ����ʽ
��3�����ݻ�ѧ����ʽ�ļ��㲽����н��⣮
��4������Ԫ�������غ���м���
����⣺��1��ϡ��ǰ�����ʵ��������䣬����Ũ���������Ϊx
250g×19.6%�Tx×98%
x=50g
��2�������������ᷴӦ������������ˮ���ʻ�ѧ����ʽΪ��Al2O3+3H2SO4�TAl2��SO4��3+3H2O
��3����������������Ϊx������Al2��SO4��3����Ϊy
Al2O3 +3H2SO4�TAl2��SO4��3+3H2O
102 294 342
x 150g×19.6% y


x�T10.2g y=34.2g
�ڸ�������������������������Ϊ�� ×100%=60%��
×100%=60%��
�۷�Ӧ��������Һ�м���39.8gˮ����������Һ��������������Ϊ�� ×100%=17.1%��
×100%=17.1%��
��4��������������Ϊz
z× =108t×95%÷��1-5%��
=108t×95%÷��1-5%��
z=204t
��Ҫ���������������Ϊ��204t÷60%=340t
�ʴ�Ϊ����1��50
��2��Al2O3+3H2SO4=Al2��SO4��3+3H2O
��3���� ��60% ��17.1%
��60% ��17.1%
��4��340
����������ǣ���˻�ѧ�еĶ��ֻ�ѧ���㣬�ۺ��Ժ�ǿ�������ѶȺܴ��ʺϻ���֪ʶ����ϰ��
��2���������������ᷴӦ�����κ�ˮд����ѧ����ʽ
��3�����ݻ�ѧ����ʽ�ļ��㲽����н��⣮
��4������Ԫ�������غ���м���
����⣺��1��ϡ��ǰ�����ʵ��������䣬����Ũ���������Ϊx
250g×19.6%�Tx×98%
x=50g
��2�������������ᷴӦ������������ˮ���ʻ�ѧ����ʽΪ��Al2O3+3H2SO4�TAl2��SO4��3+3H2O
��3����������������Ϊx������Al2��SO4��3����Ϊy
Al2O3 +3H2SO4�TAl2��SO4��3+3H2O
102 294 342
x 150g×19.6% y


x�T10.2g y=34.2g
�ڸ�������������������������Ϊ��
 ×100%=60%��
×100%=60%���۷�Ӧ��������Һ�м���39.8gˮ����������Һ��������������Ϊ��
 ×100%=17.1%��
×100%=17.1%����4��������������Ϊz
z×
 =108t×95%÷��1-5%��
=108t×95%÷��1-5%��z=204t
��Ҫ���������������Ϊ��204t÷60%=340t
�ʴ�Ϊ����1��50
��2��Al2O3+3H2SO4=Al2��SO4��3+3H2O
��3����
 ��60% ��17.1%
��60% ��17.1% ��4��340
����������ǣ���˻�ѧ�еĶ��ֻ�ѧ���㣬�ۺ��Ժ�ǿ�������ѶȺܴ��ʺϻ���֪ʶ����ϰ��

��ϰ��ϵ�д�
 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
�����Ŀ
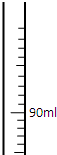 ij����ұ���������������ʲ�����ˮ��Ҳ�����ᷴӦ�����������ⶨ����������������������������ش��������⣺
ij����ұ���������������ʲ�����ˮ��Ҳ�����ᷴӦ�����������ⶨ����������������������������ش��������⣺ ij����ұ���������������������⣬����ɷֲ�����ˮҲ�����ᷴӦ��ұ�������������ⶨ����������������������������ش��������⣺
ij����ұ���������������������⣬����ɷֲ�����ˮҲ�����ᷴӦ��ұ�������������ⶨ����������������������������ش��������⣺ ij����ұ���������������������⣬����ɷֲ�����ˮҲ�����ᷴӦ��ұ�������������ⶨ����������������������������ش��������⣺
ij����ұ���������������������⣬����ɷֲ�����ˮҲ�����ᷴӦ��ұ�������������ⶨ����������������������������ش��������⣺