题目内容
5.小英同学为了测定某地区石灰石样品中碳酸钙的质量分数,取该样品15g,现将75mL的稀盐酸分三次加入石灰石样品中,每次充分反应后测得生成气体的质量,实验数据如下表:| 实验 | 第一次 | 第二次 | 第三次 |
| 加入稀盐酸的量/mL | 25 | 25 | 25 |
| 生成气体的质量/g | 2.2 | m | 1.1 |
(1)m的值是2.2g.
(2)求该样品中碳酸钙的质量分数和所用稀盐酸的质量分数(保留一位小数).
分析 (1)根据记录数据可发现,第三次实验中加入盐酸还有二氧化碳放出,并且第三次实验中只放出1.1g二氧化碳,说明盐酸有剩余,所以在第三次实验中碳酸钙已经反应完,说明第一次和第二次实验中盐酸全部反应,放出二氧化碳的质量均为2.2g进行分析;
(2)根据二氧化碳的质量求出该石灰石样品中碳酸钙的质量和参加反应的氯化氢的质量,进而求出该石灰石样品中碳酸钙的质量分数和稀盐酸的质量分数进行分析.
解答 解:(1)根据记录数据可发现,第三次实验中加入盐酸还有二氧化碳放出,并且第三次实验中只放出1.1g二氧化碳,说明盐酸有剩余,所以在第三次实验中碳酸钙已经反应完,说明第一次和第二次实验中盐酸全部反应,放出二氧化碳的质量均为2.2g,所以m的值为2.2,共产生CO2的质量=2.2g+2.2g+1.1g=5.5g;
(2)设石灰石中CaCO3的质量为x,
CaCO3+2HCl=CaCl2+H2O+CO2↑
100 44
x 5.5g
$\frac{100}{x}$=$\frac{44}{5.5g}$
x=12.5g
石灰石样品中CaCO3的质量分数为:$\frac{12.5g}{15g}$×100%=83.3%.
设所用稀盐酸的质量分数为y
CaCO3+2HCl=CaCl2+H2O+CO2↑
73 44
y×25g 2.2g
$\frac{73}{y×25g}$=$\frac{44}{2.2g}$
y=14.6%
故答案为:(1)2.2;
(2)83.3%,14.6%.
点评 本题主要考查了学生分析图表数据,解决问题的能力,以及据方程式计算的能力.

| A. | 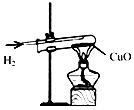 氢气还原氧化铜 氢气还原氧化铜 | B. | 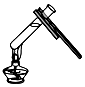 加热液体 | C. | 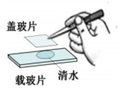 盖盖玻片 | D. |  使用测电笔 |
| 实验次数 | 金属质量(g) | 金属氧化物质量(g) |
| l | 45.0 | 84.9 |
| 2 | 45.0 | 85.1 |
| 3 | 45.0 | 85.0 |
| 平均值 | 45.0 | m |
(2)已知R的化合价为+3价,则R的相对原子质量为27.
(3)若用该金属与稀硫酸反应制取0.6克氢气,至少需要质量分数为10%的稀硫酸溶液多少克?
| 选项 | 物质(括号内为杂质) | 去除杂质的方法 |
| A | NaCl(Na2CO3) | 加入适量的Ca(OH)2溶液、过滤 |
| B | CaO(CaCO3) | 加足量的水、过滤 |
| C | CaCl2(HCl) | 加足量CaCO3、过滤 |
| D | H2SO4(HCl) | 加入适量BaCl2溶液、过滤 |
| A. | A | B. | B | C. | C | D. | D |
| 物 质 | 选用试剂(适量) | 操作方法 | |
| A | Cu(CuO) | 氧气 | 通入氧气并加热 |
| B | CO2(HCl) | 氢氧化钠溶液 | 气体通过盛有氢氧化钠溶液的洗气瓶 |
| C | NaOH(Na2CO3) | 稀盐酸 | 加入稀盐酸充分反应 |
| D | KCl溶液(K2CO3) | 氯化钙溶液 | 加入氯化钙溶液充分反应后过滤 |
| A. | A | B. | B | C. | C | D. | D |
