��Ŀ����
20���ҹ��Ƽҵ������--��°�̽�������ˡ������Ƽ���������������漰����Ҫ��ѧ��Ӧ���£���NH3+CO2+X=NH4HCO3��NH4HCO3+NaCl=NH4Cl+NaHCO3�� ��2NaHCO3$\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2��

��ش��������⣺
��1����Ӧ����X��ѧʽΪH2O��
��2����ȥ����Na2CO3��ĩ��������NaHCO3�ķ����ǽ��������ȣ�
��3������������Ӧ�к���3�ֻ�����Ӧ���ͣ�
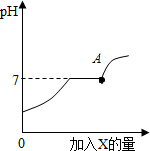
��4��������������ȼ�յ��۹�����ͼ��ʾ����A��B��C��D�仯ѧ������֮��Ϊ4��3��2��6��
���� ��1�����������غ㶨�ɷ���x��
��2������NaHCO3�����ֽ�����ش�
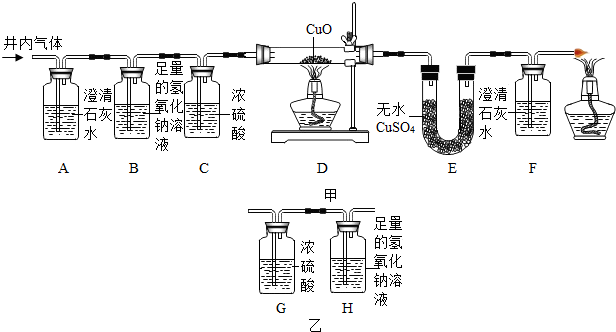
��3�����ݾ����漰���Ļ�ѧ��Ӧ��������
��4���۲찱ȼ�յ��۹���ͼ��������Ӧ������д����Ӧ�Ļ�ѧ����ʽ��������������ж��йص����⣮
��� �⣺��1�����ݻ�ѧ��Ӧǰ��ԭ���������Ŀ���䣬��Ӧǰ��������N��H��C��OԪ�ص�ԭ�Ӹ���Ϊ��N 1��H 3��C 1��O 2������Ӧ��������N��H��C��OԪ�ص�ԭ�Ӹ���Ϊ��N 1��H 5��C 1��O 3����Ӧ��X��һ�������к���2��Hԭ�Ӻ�1��Oԭ�����ԣ�xΪH2O�����H2O��
��2������NaHCO3�����ֽ��̼���ơ�ˮ�Ͷ�����̼�����ԣ���ȥ����Na2CO3��ĩ��������NaHCO3�ķ����ǽ��������ȣ�������������ȣ�
��3����NH3+CO2+X=NH4HCO3���ڻ��Ϸ�Ӧ����NH4HCO3+NaCl=NH4Cl+NaHCO3�������ڸ��ֽⷴӦ����2NaHCO3$\frac{\underline{\;\;��\;\;}}{\;}$Na2CO3+H2O+CO2�����ڷֽⷴӦ���������ֻ�����Ӧ���ͣ����3��
��4����Ӧ�Ļ�ѧ����ʽΪ4NH3+3O2$\frac{\underline{\;��ȼ\;}}{\;}$2N2+6H2O�����4��3��2��6��
���� ���⿼�������غ㶨�ɺ���ȡ������Ϣ�ó��������ʲ�������г��ӣ��Լ���ѧ��Ӧ�������͵ķ������жϣ�

| ʳ�� | ���ס����Źǡ����Ρ����⡢���������� |
| ���� | ʳ�Ρ����桢���ǡ�ʳ�ס�ζ�������͡�ʳ���� |
| ��Ʒ | �������������롢���ӡ����� |
��2��Ұ����У���һͬѧ���۷��������ˣ��۷䶾Һ�����ԣ�����ͬѧ�������÷���ˮΪ��ͿĨ���������֢״�����ᣬ������Ϊ����ˮ�ʼ��ԣ�
��3��Ұ��������С�ƴ������л�Ϥ��ijƷ�ƽ���ÿ100mL����Ӫ�����ı���������0.2g����Ӫ��������Ԫ����������Ϊ12.5%���ɴ˿ɼ���һƿ448mL��Ʒ�������������൱�ڶ��ٿ�������������������Ԫ��������

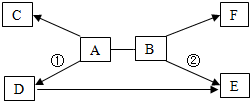 A��F�dz��л�ѧ�������������ʣ�����֮��Ĺ�ϵ��ͼ��ʾ����������ʾ���ʼ��ת����ϵ����-����ʾ�������������Ӧ�����ַ�Ӧ������P��Ӧ����δ�������
A��F�dz��л�ѧ�������������ʣ�����֮��Ĺ�ϵ��ͼ��ʾ����������ʾ���ʼ��ת����ϵ����-����ʾ�������������Ӧ�����ַ�Ӧ������P��Ӧ����δ�������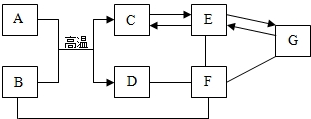 ���м��ֳ������ʼ�ķ�Ӧ��ת����ϵ��ͼ��ʾ������B��һ�ֺ�ɫ�����ĩ����ͼ�����˻�����F�����ֲ�ͬ��ѧ���ʣ������ݴ�ͼ����������⣮
���м��ֳ������ʼ�ķ�Ӧ��ת����ϵ��ͼ��ʾ������B��һ�ֺ�ɫ�����ĩ����ͼ�����˻�����F�����ֲ�ͬ��ѧ���ʣ������ݴ�ͼ����������⣮