��Ŀ����
18��ѧϰ�ˡ���������ͼ��ijС��ͬѧ���кͷ�Ӧ��һ��̽����ͬѧ�Ƿֱ�ⶨ��ϡH2SO4��NaOH��Һ��pH��20�棩������NaOH��Һ��pH����7������ڡ���С�ڡ����ڡ�������һ������NaOH��Һ����ε���H2SO4����Ӧ�Ļ�ѧ����ʽΪH2SO4+2NaOH�TNa2SO4+2H2O����ͼ2������pH������ʵʱ���ϡH2SO4��NaOH��Һ��Ӧ�����л��Һ��pH�ı仯�������������������Ƴ��˻��Һ��pH�仯�����ߣ�R����ʾ�����Һ��pHС��7��˵����ʱϡH2SO4��������̽�������У�ͬѧ��ѡ��pH�ƶ�����pH��ֽ���ⶨ��Һ��pH������Ϊ��������ԭ����pH��ʹ�÷��㣬����ʱ�����Һ��pH��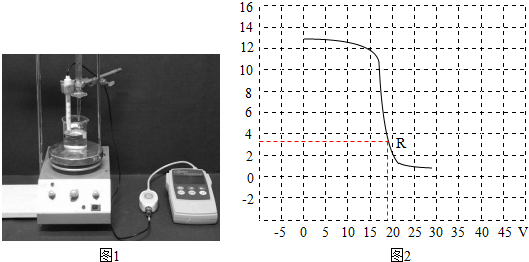
���� ��������������Һ���ڼ�Լ��ԣ�pHֵ����7��ϡ������������Ʒ�Ӧ���������ƺ�ˮ��ͼ���е�pHֵ��4��������Һ�����ԣ�ϡ������������pH��ֽ��pH�Ƶ�ʹ�÷������з�����
��� �⣺����������Һ���ڼ�Լ��ԣ�pHֵ����7��ϡ������������Ʒ�Ӧ���������ƺ�ˮ����ѧ����ʽΪ��H2SO4+2NaOH�TNa2SO4+2H2O��ͼ���е�pHֵ��4��������Һ�����ԣ�ϡ�����������̽�������У�ͬѧ��ѡ��pH�ƶ�����pH��ֽ���ⶨ��Һ��pH����������ԭ���ǣ�pH��ʹ�÷��㣬����ʱ�����Һ��pH��
�ʴ�Ϊ�����ڣ�H2SO4+2NaOH�TNa2SO4+2H2O��ϡH2SO4��pH��ʹ�÷��㣬����ʱ�����Һ��pH��
���� �������ʵ�飬��ѧ�ؽ���ʵ�顢����ʵ�飬�ǵó���ȷʵ����۵�ǰ�ᣬ���Ҫѧ�����ʵ�顢����ʵ�顢����ʵ�飬Ϊѧ�û�ѧ֪ʶ�춨������

��ϰ��ϵ�д�
 �±�Сѧ��Ԫ�Բ���ϵ�д�
�±�Сѧ��Ԫ�Բ���ϵ�д� �ִʾ��ƪϵ�д�
�ִʾ��ƪϵ�д�
�����Ŀ
9�������е����尴��������ƣ���ռ���������ǣ�������
| A�� | ���� | B�� | ������̼ | C�� | ���� | D�� | ˮ���� |
13�����ж�ʵ�������������������ǣ�������
| A�� | þ���ڿ����о���ȼ�գ�����ҫ�۰� | |
| B�� | ���ڿ�����ȼ�ղ������ĵ���ɫ���棬��������ɫ�д̼�����ζ������ | |
| C�� | ����ð״��еμ���ɫʯ����Һ����Һ��ɺ�ɫ | |
| D�� | �����ڿ�����ȼ�ղ��������İ�ɫ���� |
17�����ӵĶ�Һ�к������ᣬ�����˱����Ժ�ʹ�����̣�����ͿĨ�����������ʽ��ͣ�������
| A�� | ʳ�� | B�� | ʳ����Һ | C�� | ����ˮ | D�� | �ƾ� |
 ��Ƕ���ʶ���ʣ��ܰ������Ǹ�ȫ���˽��������磮�������Ͷ�����̼Ϊ�����ش��������⣺
��Ƕ���ʶ���ʣ��ܰ������Ǹ�ȫ���˽��������磮�������Ͷ�����̼Ϊ�����ش��������⣺