��Ŀ����
�ƾ��ֳ�Ϊ�Ҵ���������������������������Ҳ��һ�����ȼ�ϡ��õ��������Ҵ��Ļ�ѧ��Ӧ����ʽ�ǣ�(C6H10O5)n��nH2O  2nCH3CH2OH��2nCO2��
2nCH3CH2OH��2nCO2��
[ע��(C6H10O5)n����Է���������162n ]�����㣨�������1λС������
��1��CH3CH2OH����Է��������� ��
��2��100�K���������Ͽ����Ƶ��Ҵ��������Ƕ��٣�
��3���������Ƽݳ��ı�������ѪҺ���Ҵ���������80mg/L����������ѪҺռ�������ص�10%������ѪҺ���ܶ��ǣ�1kg/L������

�٣�һ������Ϊ60kg�ļ�ʻԱѪҺ�������Ҵ������������ܳ������٣�
�ڣ����н���൱����������Ϊ50%�İƵ������Ƕ��٣�
��1��46
��2���⣺��100kg���������Ͽ����Ƶ��Ҵ���������x
��C6H10O5��n+2NH2O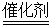 2NCH3CH2OH+2NCO2��
2NCH3CH2OH+2NCO2��
162n 92n
100kg x
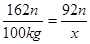
x��56.8kg
��3���⣺����һ������Ϊ60kg�ļ�ʻԱ��ѪҺ�о����������ܳ�������Ϊy
y=(60kg��10%��1kg/L)��80mg/L
=480mg
���൱����������50%�İƵ�����Ϊz
Z=480mg��50%
=960mg
����:��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� 2nCH3CH2OH��2nCO2��
2nCH3CH2OH��2nCO2��
 2nCH3CH2OH��2nCO2��
2nCH3CH2OH��2nCO2��
 2nCH3CH2OH+2nCO2��[ע��(C6H10O5)n����Է���������162n ]�����㣨�������1λС������
2nCH3CH2OH+2nCO2��[ע��(C6H10O5)n����Է���������162n ]�����㣨�������1λС������